Single-stage revision for chronic periprosthetic joint infection of the knee: a minimum 4-year follow-up
Introduction
Periprosthetic joint infection (PJI) is a challenging and complex complication that threatens both life and function. The infection rate after total knee arthroplasty (TKA) ranges from 1% to 2% (1). PJI of the knee is one of the main reasons for revision surgery in China (2).
Infected TKA is most commonly treated via two-stage revision surgery, which is the current gold standard; however, this is a costly process that requires prolonged hospitalization and two operations. Furthermore, the interval between the two operations is often associated with impaired mobility, joint stiffness, pain, and even mortality. Development of arthrofibrosis can make reimplantation difficult.
Single-stage revision for chronic PJI involves removal of the prosthesis, followed by debridement and immediate reimplantation of a new prosthesis with antibiotic-loaded cement; initial studies report a success rate of 73% (3). Single-stage revision requires only one operation, which is presumably accompanied by the inherent advantages of less morbidity, lower cost, and faster restoration of functionality.
In our center, all patients with chronic PJI after TKA that involved MRSA (methicillin-resistant Staphylococcus aureus), MRSE (methicillin-resistant Staphylococcus epidermis), fungi, and gram-negative bacilli were treated by single-stage revision. However, many studies report a lower rate of infection control using single-stage revision compared with two-stage revision. We investigated the outcome of chronic PJI patients treated via single-stage revision in terms of: (I) infection control rate; (II) functional outcome.
Methods
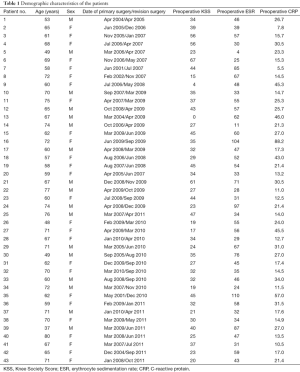
We retrospectively reviewed the records of 43 patients (43 knees) who underwent single-stage revision for chronic PJI of the knee between April 2005 and October 2011 (Table 1). We routinely used single-stage revision to treat PJI in cases of chronically infected TKA where the patient was able to tolerate anesthesia and surgery, therefore a consecutive series of 43 patients were achieved in the present review.
Full table
Diagnosis of joint infection
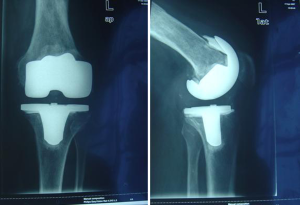
All 43 patients had been diagnosed with chronic infections of greater than 3-month duration and septic loosening of components (Figure 1). Joint aspiration was performed preoperatively in all patients. Joint aspiration protocol included: (I) cessation of antibiotics at least 2 weeks before aspiration; (II) aspiration conducted in an operating theatre; (III) no local anesthetic; (IV) if no fluid was obtained at the time of attempted aspiration, a second attempt was made intraoperatively under direct visualization of purulence; (V) if purulence was not obtained intraoperatively, periprosthetic membrane was soaked in saline for culture. The synovial fluid was sent for analysis of white blood cell count and neutrophil percentage; the sample was also cultured for aerobic and anaerobic bacteria and fungi, and incubated for 14 days (4,5). Diagnosis of infection was confirmed by pathohistological testing that demonstrated WBC >10/HPF. Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) aided in diagnosis, and were important for postoperative monitoring. Criteria for successful infection control and duration of oral antibiotics were based on constantly decreasing ESR and CRP levels, no clinical signs of infection, no further surgery with the diagnosis of PJI, and no further positive cultures after the one-stage septic exchange.
Surgical technique
All surgeries were performed by one senior surgeon (initials redacted for blinded review) experienced in knee surgery and infection treatment. Major principles of the surgical technique included: (I) original skin scar resection and medial patella approach; (II) patella eversion was not mandatory, but temporary pin fixation of the tibial tubercle was done intraoperatively to prevent patellar tendon avulsion; (III) Gigli saw and narrow straight osteotomes were applied as explant metal works to retain bone stock; (IV) cement was removed using special cement chisels, long rongeurs, curetting instruments, long drills, cement taps, and ball-headed reamers (all 43 knees were implanted with cemented components).
The procedure was separated into two stages. The first stage started with thorough and aggressive debridement and cautious removal of foreign material, components, and cement, including necrotic and inactive scar tissue (Figure 2). At least three isolated samples were collected and sent for microbiological examination and culture. The surgical area was washed with hydrogen peroxide, then soaked in aqueous betadine for at least 10 minutes. In the second stage, the patient was redraped and the surgical team rescrubbed and put on new gowns; new instruments were introduced after further debridement and another copious pulsatile lavage of the surgical area and intramedullary canals. The new prosthesis was implanted with antibiotic-loaded cement. A drainage tube was placed and retained for 3–4 days; the drain was removed when drainage fluid was less than 50 mL/day.
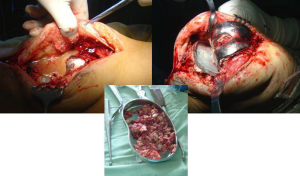
Reconstruction and prosthesis selection
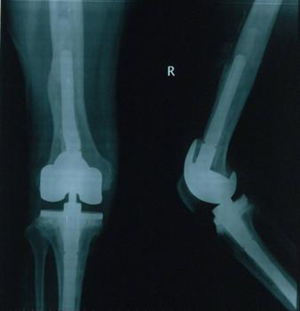
Ligamentous laxity was seen in all patients after debridement; however, there were no cases of ligament loss. Modular prostheses with stems were used in all patients without any hinge application. The Zimmer NexGen LCCK revision system (Zimmer Orthopaedics, Warsaw, IN) was used in 41 of 43 knees (95%); standard posterior-stabilized polyethylene inserts were implanted in 12 knees (28%), constrained articulating surfaces were used in 29 (67%) (Figures 3-5), and TC3 prostheses with standard inserts were used in the remaining two (5%). The implants were cemented up to the metaphyseal region with pins that were fixed onto the residual host bone to fill major bone defects. One patient (2%) had a combination of structural bone graft with pin fixation and antibiotic cement to fill a large metaphyseal defect of the femur. All allograft bone was freeze-dried, then impregnated with gentamicin before grafting. A rectus snip was made in four patients with contracture of the extensor mechanism.
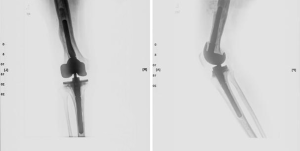
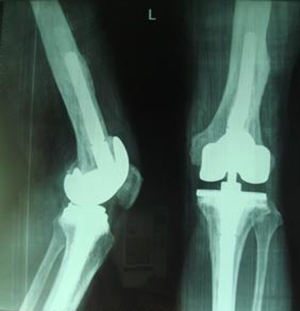
Postoperative protocols
Antibiotics were chosen based on the culture and sensitivity results and added to the cement, up to a maximum of 10% antibiotic admixture per package of cement. Antibiotic-loaded commercial cement was routinely used. Palacos R+ G 40-g cement containing 0.5-g gentamicin impregnated with 1-g vancomycin was used in 21 patients.
Appropriate intravenous antibiotics were given for 10–14 days, based on the patients’ clinical signs, ESR and CRP levels. Oral antibiotics were continued for 6 weeks; the most frequently used oral antibiotics were rifampicin (39 patients), levofloxacin (31 patients), or rifampicin and levofloxacin combined (31 patients). Two knees were infected with MRSA, one with MRSE, three with fungi, and two with gram-negative bacilli; these were treated with direct intra-articular sensitive antibiotics (vancomycin or fluconazole) after single-stage revision. Seven patients with negative synovial fluid cultures were given intravenous vancomycin and oral levofloxacin.
Postoperative rehabilitation after revision surgery differed from that after primary TKA; weightbearing with crutches was allowed as early as could be tolerated, and early mobilization was encouraged after drainage tube removal. Early continuous passive motion without angular restriction was conducted in 39 patients; mobilization was delayed in four patients because of vastus reconstruction.
Assessment of knee function
We evaluated functional outcomes using the Knee Society Score (KSS) and compared the preoperative KSS to the KSS at last follow-up.
Results
This study included 16 female and 27 male patients. The mean age was 65 years (range, 37–80 years). Demographic characteristics of the patients are shown in Table 1. The most recent follow-up was in October 2015, and the minimum duration of follow-up was 4 years (range, 4–10 years). No patient was lost before the minimum follow-up of this study. 42 patients (97%) had important comorbidities: 14 (33%) had Type 2 diabetes mellitus, 10 (23%) were morbidly obese, 13 (30%) had a previous history of debridement with administration of intravenous antibiotics, and 2 (5%) had undergone septic revision surgery.
No repeat revision surgery was necessary in 97% of 43 patients over a minimum of 4 years’ follow-up. 41 of 43 patients (95%) were free of any knee infection at last follow-up; the 4-year survival rate for being free of any knee infection was 95%. Forty-two of 43 patients (97%) were healed after the index infection without any further surgical intervention; the 4-year survival rate for being healed of the index knee infection after successful one-stage exchange was 97%.
One patient died from an unrelated reason with an asymptomatic knee, but still fulfilled the minimum 4-year follow-up of this study. One patient developed a recurrent infection for which further surgery was required. The recurrence of infection was eradicated by prosthesis removal and two-stage revision. One patient was reinfected by a new organism 1 month after revision. Fungus was cultured from synovial fluid obtained after 2 weeks without antibiotics. This was treated without surgical intervention; the patient was administered 14-day intravenous vancomycin and 15 direct intra-articular fluconazole injections over 30 days. Oral fluconazole was then administered for 3 months. Eradication of the new PJI was confirmed at the 6-month follow-up. Both these patients had had a septic prosthesis for several years and had undergone multiple debridements.
The most frequently detected bacteria in this series were methicillin-sensitive S. epidermidis and methicillin-sensitive S. aureus (Table 2). Gram-positive bacteria were still the major pathogens involved in PJI (Table 2). We applied our single-stage revision protocol for PJI due to MRSA, MRSE, fungus and gram-negative bacilli. Seven patients (16%) with negative cultures were formally diagnosed as having chronic PJI by postoperative pathohistological examination.

Full table
The inflammatory markers remained high out of normal range in nine patients (21%) despite 3 months of oral antibiotics; however, ESR and CRP levels had constantly decreased and synovial tests were negative. After 6 months of oral antibiotics, the inflammatory markers of all nine patients had reduced back to normal range.
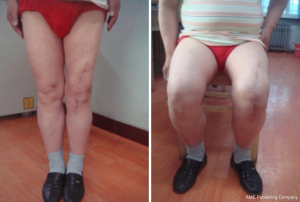
The knee range of motion improved significantly from a mean value of 50° (± 25.6 SD; range, 8°–144°) to 76° (± 33 SD; range, 8°–144°). The results for pain (maximum of 30 points) were a mean preoperative 4.7 (± 8.6; range, 0–25) and improved significantly postoperatively to 17.9 points (± 10.6; range, 0–30). A significant improvement was also found regarding the function pre- and postoperatively (Figure 6). Preoperatively, the mean KSS score was 33.6 (range, 0–61) points. At the most recent follow-up, the mean KSS score had increased significantly to 63.9 (range, 43–82) points.
Discussion
By December 2015, we had treated 98 chronic knee PJIs by single-stage revision. In short-term follow-up cases, the infection rate might increase in the future owing to additional new infections as the median time to reinfection might be as high as 4 years (6); therefore patients between April 2005 and October 2011 with a minimum of 4 years’ follow-up were selected for this study. This gave us a consecutive series of 43 patients, which is higher than in most comparable studies (7-11).
The reported success of two-stage exchange arthroplasty varies, with some studies showing a 100% rate of infection control (12-15). A review of PJIs in patients who underwent resection arthroplasty, spacer insertion and reimplantation, reported that reimplantation occurred in 82.7% of 504 cases, and the overall success rate with a minimum 1-year follow-up was 53.1% (16).
A recent systematic review of the literature (17) retrieved only four single-stage studies (3,8-10), some of which were limited to fewer than 20 TKAs and only one of which was published in the last decade (8). The study with the largest number of TKAs [104] available for follow-up reported a rate of infection control of 73% after one-stage exchange (3). A multicentric retrospective study comparing single- and two-stage revisions for infection found no difference between techniques in eradicating infection (7).
The infection control rate of our single-stage revision was 95% (41 of 43 patients). Residual biofilm and necrotic tissues were the main reason for infection recurrence. Biofilm produced by bacteria forms between 36 hours and 3 weeks (18), preventing antimicrobial agents from penetrating through to the underlying bacteria. Therefore, rigorous removal of all foreign materials and aggressive debridement of inflamed and necrotic scar tissues, including a total synovectomy, are essential for septic revision (19). Final debridement of bone and posterior soft tissues must be as radical as possible, and include areas of osteolysis and non-viable bone.
Aggressive debridement and removal of components generally led to ligamentous instability and bone loss, resulting in mismatch of the flexion and extension gap. Therefore, modular prostheses with stems were implanted in all patients in this study. Mixing antibiotics into the cement impacts the quality of the cement; hence, only a maximum of 10% of the total cement amount should be antibiotic powder (20). The level of released antibiotic is reportedly sufficient for at least 4 months (21-24).
Over the last two decades the prevalence of infections caused by organisms such as MRSA and MRSE has increased, mainly owing to inappropriate use of antibiotics (25). Common methods of treatment include debridement, antibiotic-impregnated spacer, long-term intravenous antibiotics, and revision surgery, which has a high failure rate ranging from 24% to 82% despite aggressive antibiotic treatment (14,26-28). We successfully treated two MRSA, one MRSE, three fungal, and two gram-negative bacilli infections with direct intra-articular sensitive antibiotics (vancomycin or fluconazole) after single-stage revision. A previous study reported excellent infection control in 18 patients with MRSA-infected knees treated by single-stage revision and direct intra-articular vancomycin injection (29). Another study of 14 patients with fungal PJI treated using single-stage revision revealed an acceptable rate of a satisfactory outcome (30).
Isolation of the infecting organism from cultures of fluid or tissue obtained from the affected joint is critical for the selection of appropriate antibiotic therapy and providing insight into prognosis. Any use of antibiotics in the 2 weeks before obtaining culture samples is associated with a lower yield of cultures (31). In chronic PJI, most microorganisms exist in a biofilm attached to the implant surface and surrounding tissues (32); hence, failure of tissue cultures to recover infecting organisms encapsulated in a biofilm leads to negative culture results. After April 2009, our institution incubated synovial fluid samples in blood culture flasks, which reportedly improves the detection of microorganisms and decreases contamination compared with conventional agar or broth methods (5,33). The overall incidence of negative culture in our study was 16% (7/43); however, the incidence of negative culture before March 2009 was 38% (5/13). After April 2009, the incidence of negative culture in our institution was only 7% (2/30).
The majority of PJIs (65–90%) are caused by common aerobic gram-positive microorganisms (34,35), with most identified as Staphylococcus species that can be effectively treated with parenteral vancomycin. Our higher infection control rates with intravenous and topical injection of vancomycin for negative culture PJI suggest that vancomycin-sensitive gram-positive organisms may still be the most common culprit in culture-negative infections.
Increased exposure time is associated with high risk of surgical field infection (36). Therefore, the experience of the surgical team and availability of special instruments for the explant of components and cement are important in shortening the duration of surgery. Preparation of surgical instruments and recruitment of the surgical team are vital in preoperative planning of single-stage revision.
The infection rate is significantly increased in cases with a history of previous surgical procedures (37), and peripheral vascular disease is another known risk factor for PJI (38). The two patients in our series who had a recurrence of infection had both undergone multiple debridements at other institutions and both had poor peripheral vascular condition. Poor peripheral vascular condition could potentially predispose to persistence of infection either by impaired local immune response due to decreased oxygen tension or by impaired delivery of systemic antibiotics at the site of infection. In future, PJI with a history of multiple debridements will be considered an absolute exclusion criterion for single-stage revision.
Our study had some limitations. First, it was an observational cohort study without any control group. Second, although the number of included patients was higher than in most other studies reporting single-stage revision TKA after PJI and no patient was lost to follow-up, the number of patients was too small to perform an adequately powered analysis of prognostic factors. Third, the minimum follow-up was 4 years, and the infection rate might increase in the future due to additional new infections.
Conclusions
We reviewed 43 patients with PJI after TKA treated by single-stage revision with a minimum 4-year follow-up. Single-stage revision achieved a high rate of infection control (95%) and an acceptable functional outcome (mean KSS 63.9).
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2017.06.05). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by institutional/regional/national ethics committee/ethics board of First Teaching Hospital of Xinjiang Medical University (No. 20150923-02) and written informed consent was obtained from all patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Phillips JE, Crane TP, Noy M, et al. The incidence of deep prosthetic infections in a specialist orthopaedic hospital: a 15-year prospective survey. J Bone Joint Surg Br 2006;88:943-8. [Crossref] [PubMed]
- Feng B, Weng X, Lin J. Postoperative complications and revision surgery following primary total knee arthroplasty after midterm follow-up. Zhonghua Wai Ke Za Zhi 2015;53:106-9. [PubMed]
- von Foerster G, Klüber D, Käbler U. Mid- to long-term results after treatment of 118 cases of periprosthetic infections after knee joint replacement using one-stage exchange surgery. Orthopade 1991;20:244-52. [PubMed]
- Fink B, Makowiak C, Fuerst M, et al. The value of synovial biopsy, joint biopsy, joint aspiration and C-reactive protein in the diagnosis of late peri-prosthetic infection of total knee replacements. J Bone Joint Surg Br 2008;90:874-8. [Crossref] [PubMed]
- Schäfer P, Fink B, Sandow D, et al. Prolonged bacterial culture to identify late periprosthetic joint infection: a promising strategy. Clin Infect Dis 2008;47:1403-09. [Crossref] [PubMed]
- Kubista B, Hartzler RU, Wood CM, et al. Reinfection after two-stage revision for periprosthetic infection of total knee arthroplasty. Int Orthop 2012;36:65-71. [Crossref] [PubMed]
- Bauer T, Piriou P, Lhotellier L, et al. Results of reimplantation for infected total knee arthroplasty: 107 cases. Rev Chir Orthop Reparatrice Appar Mot 2006;92:692-700. [Crossref] [PubMed]
- Buechel FF, Femino FP, D’Alessio J. Primary exchange revision arthroplasty for infected total knee replacement: a long term study. Am J Orthop 2004;33:190-8. [PubMed]
- Göksan SB, Freeman MA. One-stage reimplantation for infected total knee arthroplasty. J Bone Joint Surg Br 1992;74:78-82. [PubMed]
- Lu H, Kou B, Lin J. One-stage reimplantation for the salvage of total knee arthroplasty complicated by infection. Chin J Surg 1997;35:456-8. [PubMed]
- Scott IR, Stockley I, Getty CJ. Exchange arthroplasty for infected knee replacements: a new two-stage method. J Bone Joint Surg Br 1993;75:28-31. [PubMed]
- Hsieh PH, Shih CH, Chang YH, et al. Two-stage revision hip arthroplasty for infection: comparison between the interim use of antibiotic-loaded cement beads and a spacer prosthesis. J Bone Joint Surg Am 2004;86-A:1989-97. [Crossref] [PubMed]
- Lim SJ, Park JC, Moon YW, et al. Treatment of periprosthetic hip infection caused by resistant microorganisms using 2-stage reimplantation protocol. J Arthroplasty 2009;24:1264-69. [Crossref] [PubMed]
- Volin SJ, Hinrichs SH, Garvin KL. Two-stage reimplantation of total joint infections: a comparison of resistant and non-resistant organisms. Clin Orthop Relat Res 2004;94-100. [Crossref] [PubMed]
- Yamamoto K, Miyagawa N, Masaoka T, et al. Clinical effectiveness of antibiotic-impregnated cement spacers for the treatment of infected implants of the hip joint. J Orthop Sci 2003;8:823-8. [Crossref] [PubMed]
- Gomez MM, Tan TL, Manrique J, et al. The fate of spacers in the treatment of periprosthetic joint infection. J Bone Joint Surg Am 2015;97:1495-502. [Crossref] [PubMed]
- Insall JN, Dorr LD, Scott RD, et al. Rationale of The Knee Society clinical rating system. Clin Orthop Relat Res 1989;13-4. [PubMed]
- Bjarnsholt T, Ciofu O, Molin S, et al. Applying insights from biofilm biology to drug development—can a new approach be developed? Nat Rev Drug Discov 2013;12:791-808. [Crossref] [PubMed]
- Singer J, Merz A, Frommelt L, et al. High rate of infection control with one-stage revision of septic knee prostheses excluding MRSA and MRSE. Clin Orthop Relat Res 2012;470:1461. [Crossref] [PubMed]
- Springer BD, Lee BC, Osmon D, et al. Systemic safety dose antibiotic loaded cement spacers after resection of an infected total knee arthroplasty. Clin Orthop Relat Res 2004;47. [Crossref] [PubMed]
- Bertazzoni Minelli E, Benini A, Magnan B, et al. Release of gentamicin and vancomycin from temporary human hip spacers in two-stage revision of infected arthroplasty. J Antimicrob Chemother 2004;53:329-34. [Crossref] [PubMed]
- Fink B, Vogt S, Reinsch M, et al. Sufficient release of antibiotic by a spacer 6 weeks after implantation in two-stage revision of infected hip prostheses. Clin Orthop Relat Res 2011;469:3141-47. [Crossref] [PubMed]
- Masri BA, Duncan CP, Beauchamp CP. Long-term elution of antibiotics from bone-cement: an in vivo study using the prosthesis of antibiotic-loaded acrylic cement (PROSTALAC) system. J Arthroplasty 1998;13:331-8. [Crossref] [PubMed]
- Hsieh PH, Chang YH, Chen SH, et al. High concentration and bioactivity of vancomycin and aztreonam eluted from Simplex cement spacers in two-stage revision of infected hip implants: a study of 46 patients at an average follow-up of 107 days. J Orthop Res 2006;24:1615-21. [Crossref] [PubMed]
- Gehrke T, Parvizi J. Proceedings of the International Consensus Meeting on Periprosthetic Joint Infection. Philadelphia: Musculoskeletal Infection Society, 2013.
- Kilgus DJ, Howe DJ, Strang A. Results of periprosthetic hip and knee infections caused by resistant bacteria. Clin Orthop Relat Res 2002;116-24. [Crossref] [PubMed]
- Mittal Y, Fehring TK, Hanssen A, et al. Two-stage reimplantation for periprosthetic knee infection involving resistant organisms. J Bone Joint Surg Am 2007;89:1227-31. [Crossref] [PubMed]
- Salgado CD, Dash S, Cantey JR, et al. Higher risk of failure of methicillin-resistant Staphylococcus aureus prosthetic joint infections. Clin Orthop Relat Res 2007;48-53. [PubMed]
- Whiteside LA, Peppers M, Nayfeh TA, et al. Methicillin-resistant Staphylococcus aureus in TKA treated with revision and direct intraarticular antibiotic infusion. Clin Orthop Relat Res 2011;469:26-33. [Crossref] [PubMed]
- Klatte TO, Kendoff D, Kamath AF, et al. Single-stage revision for fungal peri-prosthetic joint infection. Bone & Joint J 2014;96-B:492. [Crossref] [PubMed]
- Zimmerli W, Trampuz A, Ochsner PE. Prosthetic joint infections. N Engl J Med 2004;351:1645. [Crossref] [PubMed]
- Gristina AG, Costerton JW. Bacterial adherence and the glycocalyx and their role in musculoskeletal infection. Orthop Clin North Am 1984;15:517-35. [PubMed]
- Hughes JG, Vetter EA, Patel R, et al. Culture with BACTEC Peds Plus/F bottle compared with conventional methods for detection of bacteria in synovial fluid. J Clin Microbiol 2001;39:4468-71. [Crossref] [PubMed]
- Marculescu CE, Berbari EF, Cockerill FR 3rd, et al. Fungi, mycobacteria, zoonotic and other organisms in prosthetic joint infection. Clin Orthop Relat Res 2006;64-72. [Crossref] [PubMed]
- Moran E, Masters S, Berendt AR, et al. Guiding empirical antibiotic therapy in orthopaedics: the microbiology of prosthetic joint infection managed by debridement, irrigation and prosthesis retention. J Infect 2007;55:1-7. [Crossref] [PubMed]
- Pulido L, Ghanem E, Joshi A, et al. Periprosthetic joint infection: the incidence, timing, and predisposing factors. Clin Orthop Relat Res 2008;466:1710-15. [Crossref] [PubMed]
- Wilson MG, Kelley K, Thornhill TS. Infection as a complication of total knee replacement arthroplasty. Risk factors and treatment in sixty-seven cases. J Bone Joint Surg Am 1990;72:878-83. [Crossref] [PubMed]
- Bozic KJ, Lau E, Kurtz S, et al. Patient-related risk factors for postoperative mortality and periprosthetic joint infection in medicare patients undergoing TKA. Clin Orthop Relat Res 2012;470:130-7. [Crossref] [PubMed]
Cite this article as: Momin M, Li G, Wang Y, Maimaitiming A, Cao L. Single-stage revision for chronic periprosthetic joint infection of the knee: a minimum 4-year follow-up. Ann Joint 2017;2:40.