Total hip arthroplasty via the direct anterior approach—off the table
Introduction
Originally the direct anterior approach was first described by Carl Heuter in 1881 (1,2). He described the interval between the tensor fascia lata and the sartorius muscles for accessing the hip joint. Later, the approach was popularized by Marius Smith-Peterson who utilized the approach for initial hip replacement surgery. Over the next decade, the approach was further developed by the Judets and Karl Keggi (3).
Several techniques and modifications have been developed for performing the direct anterior approach including utilization of an orthopaedic table, a sterile hook attachment to the table, and using a standard operating room table. This article will focus on preforming the direct anterior approach for total hip arthroplasty using a standard operating room table.
The learning curve for the direct anterior approach is steep. Early in the learning process, surgeons should select end stage osteoarthritis patients with smaller amounts of muscle mass and a valgus femoral neck shaft angle. It is also prudent to select patients without an abdominal pannus to prevent any sort of wound complications as the incision is placed on the anterior and lateral aspect of the thigh. Patients to avoid include those with previous hip surgery, acetabular protrusion, contractures, or shortening (4).
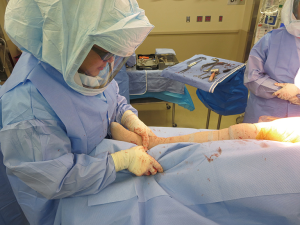
In the operating room, we prefer to give the patients spinal anesthesia but general anesthesia can be used if spinal is contraindicated. The main goal of anesthesia is to gain complete muscle relaxation. The patients are positioned supine on either a Jackson table or regular table with an arm board positioned at the level of the contralateral leg. A 2-inch bump is placed under the ASIS at a level of just above the break in the table. This allows for flexion of the foot of the table during femoral exposure. Figure 1 displays appropriate patient position on the table with relation to the break in the table. The operative leg is prepped and draped in standard orthopedic fashion. Some surgeons choose to prep and drape the contralateral leg as well to allow for better leg length evaluation intraoperatively (5).
The incision
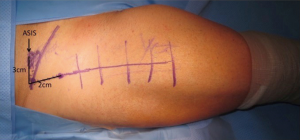
A 6–8 cm incision is placed directly over the tensor fascia lata muscle beginning approximately 2 cm distal and 2–3 cm lateral in relation to the ASIS (Figure 2). We prefer the incision to angle laterally to avoid injury to the lateral femoral cutaneous nerve.
The approach
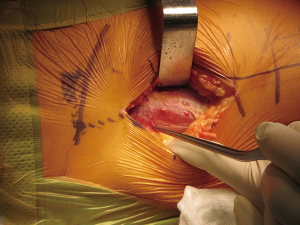
The incision is carried through the skin and subcutaneous fat. It is important to maintain meticulous hemostasis throughout the entire approach. Identify Scarpa’s fascia and incise it in line with the skin incision. Once through Scarpa’s fascia there will be a loose layer of adipose tissue superficial to the tensor fascia lata. A careful preservation of the tensor fascia lata fascial layer will allow identification of the proper interval for this approach. Four factors will identify the tensor fascia lata: (I) the muscle origin will be lateral to the ASIS; (II) the fascia will be translucent allowing the red hue of the TFL to show through; (III) perforating vessels are identified coursing into the TFL; (IV) the fascial layer will peel easily from the muscle surface when separated during dissection (Figure 3).
Blunt dissection is carried down to the pericapsular fat of the superior femoral neck and a retractor is placed extracapsularly on the superior femoral neck. A small incision in the medial TFL fascia is made and blunt dissection is carried down to the extracapsular inferior femoral neck and a second retractor is placed here. The electrocautery device is then used to incise the lateral rectus femoris fascia. It is now important to identify the lateral femoral circumflex artery and vein located in the pericapsular fat superficial to the capsule and ligate them. Once the vessels have been identified and appropriately ligated, the remainder of the pericapsular fat can be removed. An assistant will then elevate the leg to relax the femoral neurovascular bundle and the cobb elevator is advanced just superior to the capsule and below the rectus femoris and iliopsoas muscles until it advances into the retroperitoneal space (Figures 4,5). The cobb is then replaced with a retractor. The capsulotomy and/or capsulectomy is then performed and retractors are placed intracapsularly (6).

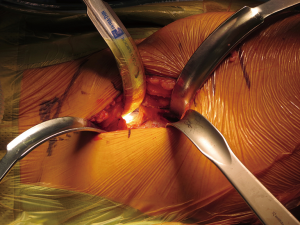
Femoral neck cut
An assistant imparts internal rotation to the femur and a cut is made approximately 1 cm above the lesser trochanter. A second neck cut is carried out approximately 1 cm proximally to first cut and this “napkin ring” is removed. A corkscrew is used to remove the femoral head.
Acetabular preparation and instrumentation
A retractor is placed medial to the transverse acetabular ligament. A second retractor is then placed laterally between the capsule and labrum junction. An incision is then made in the inferior posterior capsule and a retractor is placed in the space between the labrum and capsule. This step allows posterior retraction of the femur and greater acetabular exposure. Once retractors are placed, the periacetabular soft tissue can be resected to further develop exposure. Reaming and acetabular cup implantation can then be carried out in standard fashion.
Femoral preparation
For most surgeons, the femoral exposure and preparation is the most difficult part of the procedure. To begin the femoral release, the leg is placed into a figure-of-four position to place the inferior medial capsule on stretch. An electrocautery device is used to subperiosteally capsular release is carried out from the 6 o’clock position on the inferior medial aspect of the femoral neck cut to the 3 o’clock (right hip) or 9 o’clock (left hip) position (Figure 6).
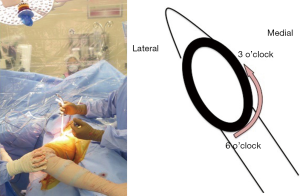
Next the retractors are removed. A retractor is then placed at the 4:30 position (right hip) or 7:30 position (left hip) and a retractor is placed at the 12:00 position superior to the greater trochanter. With one hand on the superior retractor and another hand pulling the femur laterally, the femur is brought into a more lateral position. While attempting this maneuver, the assistant is pulling longitudinal traction on the leg and externally rotating the limb. Once the femur is positioned as lateral as it can be, continue the capsular release from 3/9 o’clock to 12:00. Reposition the femur as often as needed fully clear the posterior lateral aspect of the greater trochanter from behind the acetabulum.
Once the femur is fully exposed, the contralateral limb is abducted and the operative limb is adducted. The bed is placed in slight trendelenburg and the bottom of the bed is flexed to further provide visualization. Sequential broaching is carried out in usual fashion. Again, fluoroscopy is used to provide guidance for proper stem fit and fill. Trialing of components is then performed in standard fashion with adjunct fluoroscopy as needed for assessment of leg lengths (7). Leg lengths are also assessed by measurement of medial malleoli at the end of the table (Figure 7).
Closure
Copious irrigation is preformed and the previously coagulated ascending branches of the lateral femoral circumflex arteries are assessed. Often times they will need to be re-coagulated to prevent post-operative hematoma formation. If the capsule is preserved, a running barbed suture is used to carry out closure. The tensor fascia lata fascia is then re-approximated and skin is closed. A silver impregnated silicone dressing is applied.
Aftercare
Patients are allowed to weight bear as tolerated to their operative extremity. Patients are encouraged to ambulate as needed and do not need formal physical therapy. Dressing is removed at 7 days.
Tips and tricks
Our institution has found no difference in stability when considering capsulectomy versus capsulotomy. Surgeons earlier in their learning curve may find it easier to preform capsulectomy until more comfortable with the procedure.
Insertion of acetabular reamers and components can be a difficult portion of the procedure. This can be made easier by first inserting the posterior lip of the reamer/acetabular shell and elevating one’s hand to guide the anterior aspect into the acetabulum. Additionally, the reamer head and reamer handle can be inserted into the acetabulum separately. The reamer head is inserted and positioned with a kocher clamp. Next the reamer handle is attached and reaming can be carried out. This will minimize soft tissue trauma.
Once the acetabular cup has been implanted, an osteotome can be used to remove inferior osteophytes. This eliminates inferior impingement and allows for easier femoral exposure.
To prevent a residual divot formation and slight invagination of the healed skin incision, take care to re-approximate the subcutaneous fat layer upon closure.
Pitfalls
In patients with excessive external rotation posture of their limb or those with a femoral neck fracture, the skin incision can erroneously be placed too far medially. A medially based incision greatly complicates the approach. To prevent this mistake, an assistant can hold the limb in a neutral position when planning an incision and during the approach to allow accurate anatomical navigation.
Failure to maintain meticulous hemostasis can impede visualization in this minimally invasive approach. As a result, anatomy can be obscured increasing risk for iatrogenic injury. With each layer of dissection be sure to identify and coagulate active bleeding vessels.
Haphazard placement of retractors can lead to anatomic landmark disruption and confusion during the approach. In particular, when exposing the hip capsule prior to capsulotomy, it is imperative to ensure no muscle fibers overlay the capsule to avoid injury to the femoral neurovascular bundle.
A femoral neck cut placed too far caudally can risk implant stability and potential greater trochanter injury. Take special care to identify level of femoral neck cut using fluoroscopy if necessary.
Hasty attempts at femoral broaching prior to adequate exposure can be catastrophic resulting in intraoperative fracture. Proper releases and manipulation must be carried out in order to ensure appropriate trajectory of femoral instrumentation.
Conclusions
The direct anterior approach has historically been utilized for total hip arthroplasty with a recent resurgence in popularity. The steep learning curve can be overcome with proper patient selection and meticulous technique. This article can provide guidance for the novice surgeon as well as those more experienced.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2017.08.04). ZP reports personal fees from Ortho Development, personal fees from Depuy Synthes, personal fees from Ethicon, outside the submitted work. FO reports personal fees from Stryker, outside the submitted work. AO reports personal fees from Smith and Nephew, personal fees from Stryker, outside the submitted work. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Rachbauer F, Kain MS, Leunig M. The history of the anterior approach to the hip. Orthop Clin North Am 2009;40:311-20. [Crossref] [PubMed]
- Connolly KP, Kamath AF. Direct anterior total hip arthroplasty: Literature review of variations in surgical technique. World J Orthop 2016;7:38-43. [Crossref] [PubMed]
- Judet J, Judet R. The use of an artificial femoral head for arthroplasty of the hip joint. J Bone Joint Surg Br 1950;32-B:166-73. [PubMed]
- de Steiger RN, Lorimer M, Solomon M. What is the learning curve for the anterior approach for total hip arthroplasty? Clin Orthop Relat Res 2015;473:3860-6. [Crossref] [PubMed]
- Post ZD, Orozco F, Diaz-Ledezma C, et al. Direct anterior approach for total hip arthroplasty: indications, technique, and results. J Am Acad Orthop Surg 2014;22:595-603. [Crossref] [PubMed]
- York PJ, Smarck CT, Judet T, et al. Total hip arthroplasty via the anterior approach: tips and tricks for primary and revision surgery. Int Orthop 2016;40:2041-8. [Crossref] [PubMed]
- Ji W, Stewart N. Fluoroscopy assessment during anterior minimally invasive hip replacement is more accurate than with the posterior approach. Int Orthop 2016;40:21-7. [Crossref] [PubMed]
Cite this article as: Boyle P, Post Z, Orozco F, Ong A. Total hip arthroplasty via the direct anterior approach—off the table. Ann Joint 2017;2:75.