
Arthroscopy of the pediatric hip
Introduction
The evolution of pediatric hip arthroscopy mirrors its exponential growth in adult patients (1), as significant recent advances within the field of technology and techniques have propelled the procedure forward. The first reported use of hip arthroscopy in pediatric patients came in 1977, as Gross (2) reported using arthroscopy in 28 patients to assist in the diagnosis of a variety of disorders including Legg-Calve-Perthes disease (LCPD), developmental dysplasia of the hip (DDH), and slipped capital femoral epiphysis (SCFE). Since then, there have been substantial developments in the field, leading to greatly simplified and reproducible techniques, in addition to improved treatment and clinical results. With its’ minimally invasive approach, the ability to directly visualize the articular surface, and an expedited recovery with lower rates of major complications relative to previously described open surgical techniques (3,4), it is no wonder that hip arthroscopy continues to grow in popularity.
The most common indications for hip arthroscopy in the pediatric population are similar to those seen in adults, and include hip impingement and intra-articular labral and/or chondral injuries. Femoroacetabular impingement (FAI) is a dynamic process characterized by abnormal morphology of the femoral head-neck junction (cam-type), acetabular rim (pincer-type), or a combination of the two, leading to abnormal contact (or impingement) and consequent intra-articular injury (5). While idiopathic FAI is the most common cause of hip impingement, there are also many other pediatric hip conditions that can lead to non-idiopathic impingement, which essentially includes any pathological entity that can affect the morphology or shape of the hip joint. Examples of these conditions include LCPD, SCFE, spondyloepiphyseal dysplasia (SED), trauma, and hereditary multiple exostoses (HME). A mildly incongruent joint secondary to LCPD, for example, can exhibit impingement secondary to residual deformity of the femoral head. In addition, many other non-impingement pediatric pathologies can also be treated with hip arthroscopy, and indications have expanded to the treatment of traumatic labral tears, removal of loose bodies secondary to developmental or traumatic origin, osteochondritis dissecans, tears of the ligamentum teres, chondral and morphological pathology secondary to avascular necrosis (AVN), septic arthritis and/or adjacent osteomyelitis of the hip, and the treatment of hip dysplasia.
Evaluation and decision-making
When assessing if a pediatric patient is a candidate for hip arthroscopy, it is paramount to perform a thorough medical and developmental history and physical examination to fully appreciate the underlying source of pain and/or functional limitations. On history, it is important to determine the temporality, location, quality, and duration of their symptoms. Most intra-articular hip pathologies will present with pain located in the anterior aspect of the hip joint or groin, as is demonstrated by the prototypical “C” sign (6). However, intra-articular hip pathology in the pediatric population can often present as knee pain, which is a common reason for misdiagnosis and delay in treatment (7). In trying to ascertain whether the pain is from a form of impingement or intra-articular pathology, positions and activities of discomfort should be explored. In many cases the symptoms develop insidiously, but occasionally a traumatic or mechanical episode will be a direct precipitant. Mechanical symptoms such as locking or clicking are often thought to suggest intra-articular pathologies. However, more commonly seen in the adolescent patient is coxa saltans, or snapping hip, which occurs when the iliopsoas snaps over the anterior aspect of the acetabulum (coxa saltans interna) or when the iliotibial band snaps over the greater trochanter (coxa saltans externa). These conditions are most commonly treated effectively with non-operative interventions, such as physiotherapy and activity modification (8). The patient’s developmental history is important, including assessment of risk factors for hip dysplasia, LCPD, and other early presenting pathologies. Family history can also be contributory, as many developmental hip conditions have a familial component.
The physical examination of the hip should include observation of the patient’s gait, noting signs of analgia, weakness, Trendelenburg, thrust, or other abnormalities. Palpation around the hip should be performed to evaluate for extra-articular causes of hip pain, such as iliotibial band syndrome, tendonitis, or bursitis. Range of motion (ROM) of the hip is assessed in the supine and prone positions, and limits to flexion, extension, and rotation should be documented. Clinicians should have a high clinical suspicion for other pathologies when examining a child with an externally rotated position such as is found in SCFE, or when a patient presents with a limp and limited unilateral abduction seen in LCPD and AVN. A systematic review of physical exam maneuvers for the diagnosis of FAI and labral pathology found 21 studies describing 18 different special tests (9). These include but are not limited to the anterior impingement test or FADIR (flexion, adduction and internal rotation) test, which is believed to be the most sensitive test for intra-articular hip pathology with a sensitivity approaching up to 99% (10). The FABER (flexion, abduction and external rotation) test, and the posterior impingement test are other commonly described tests for hip impingement (11). Other aspects to assess on examination include muscle bulk, atrophy, power, and a comprehensive neurovascular examination of the lower extremity. One must be careful to not omit the spine and knee exam as referred pain from either source can masquerade as hip pathology (12).
Imaging
Many imaging modalities exist to help diagnose and plan for the treatment of pediatric hip disorders, and it is important to understand the value, role, and implications for each. Standard anteroposterior (AP) and lateral radiographs of the pelvis and hips can help detect various pathologies. In the pediatric patient, we commonly begin by obtaining AP and ‘frog-leg’ lateral radiographs of the pelvis, allowing for orthogonal views of the proximal femur and providing comparison to the contra-lateral side. This is especially useful for clinicians who are less familiar with pediatric radiographs, as the physis of the proximal femur or the tri-radiate cartilage can sometimes be misinterpreted as a fracture or other pathology. The AP view is particularly important for assessing preliminary congruence of the joint, lateral acetabular coverage and depth, deformity, gross bone density, and to help rule out osteonecrosis. Depending on the pathology found or suspected, further specialized radiographic views can also be performed for diagnostic and surgical planning purposes, such as Dunn views or false profile views (13).
Advanced imaging modalities including computed tomography (CT) and magnetic resonance imaging (MRI) play an important role in the diagnosis and management of the pediatric hip, given the complex three-dimensional morphology of the joint and its’ degrees of motion. CT scans are the gold standard for assessing bony architecture and rotational profile, but their use must be judicious in the pediatric patient due to the radiation involved (14). Low dose CT options and protocols are now available, but institution-specific protocols should be discussed with musculoskeletal radiologists (15). MRI scans have become a valuable tool in the workup and diagnosis of hip pathologies and allows for visualization of the soft tissues that the CT scan does not. There is great variability in MRI scanners and protocols, thus it is important to be aware of the options and preferences with your radiologist. An appropriately protocoled MRI in a modern and powerful scanner should be able to provide an accurate assessment of the cartilage, labrum, surrounding musculature, physes, and even the vascular status of the bone. Originally, a MR arthrogram showed better results when compared to non-contrast imaging, however 3T MR has recently shown a high sensitivity for the evaluation of the labrum and cartilage in patients with clinically suspected FAI (16). This is especially encouraging in the pediatric patient, as it means that a less-invasive non-arthrogram MRI is adequate in most cases for imaging the labrum and cartilage as well as for diagnosing and ruling out other potential pathology.
Treatment
The reported clinical results of hip arthroscopy in pediatric patients have been generally positive. A case series published in 2005 examined a number of different pathologies treated with arthroscopy, reporting clinical improvement in 83% of the 54 pediatric and adolescent patients with underlying diagnoses including labral tears, LCPD, DDH, juvenile arthritis, SED, AVN, SCFE, and osteochondral fracture (17). Loose body retrieval, debridement of synovitis, and repair and microfracture of osteochondral lesions have also demonstrated favorable results with low rates of complications and improved functional outcomes (18,19).
An important element of pediatric hip arthroscopy is understanding the inherent technical limitations. Treating pediatric hip conditions arthroscopically is challenging for even the experienced hip arthroscopist as the joint is often smaller and the clinical implications of iatrogenic cartilage and labral injury, or suboptimal restoration of hip biomechanics and anatomic reconstruction, are more severe. As a result, it is imperative that the pediatric hip arthroscopist be intimately familiar with the pathology being treated and have alternative reconstructive options should they be required. For example, although there is a growing role for arthroscopic labral repair and even capsulorrhaphy in the dysplastic hip (20), pelvic osteotomies must be considered when adequate acetabular coverage is in question (21). Arthroscopy can successfully treat the intra-articular component of the deformity, however restoration of biomechanical properties may require additional osteotomies and reconstruction.
The equipment and preparation necessary for pediatric hip arthroscopy is largely the same as that required for adults. That said, the smaller patient requires special attention. Commercially available hip distraction table attachments can be too large for particularly young and small patients, In these cases a pediatric fracture table with an independent traction arm can be used as an alternative. Additionally, small feet can be a challenge with respect to foot slippage in the traction device, and planning for appropriately sized boots, liners, and reinforcements where necessary is important. Depending on the indication for surgery, one can even avoid traction altogether, such as in cases of septic arthritis where there is already a large effusion, and work in the central compartment is limited. When initially distracting the hip as the first portal is developed, it can be very helpful to do so after already using a spinal needle to help break the hip’s fluid seal. Because most young hips are quite a bit more flexible, breaking the seal of the hip using only traction alone can be somewhat more traumatic. Another element to generally consider when performing pediatric hip arthroscopy is the management of the capsule. More aggressive capsular closure/plication in younger hips with increased laxity, or concomitant hip dysplasia, may lead to an improvement in post-operative outcomes. However, even with a recent trend towards routine capsular closure, post-operative instability is rarely reported and evidence-based indications for capsular closure remain unclear (22).
Femoroacetabular impingement
The evaluation and treatment of FAI has gained significant popularity. Although the etiology of cam morphology is not completely understood, recent studies have supported a correlation between physeal closure, physical activity levels and the development of cam morphology (23-25). Agricola (26) and Carsen et al. (27), have shown in cross-sectional studies that significant increases in cam deformity at the head neck junction only occurred in pediatric patients with an open physis over the course of physeal closure. These results support the notion that cam morphology is likely developed during, and not after, the closure of the physis and can be affected by certain aggravating sports, specifically, hockey, soccer, basketball and American football (23-26). However, symptomatic FAI is not as straight forward as meeting a certain morphological angular threshold. A retrospective study of amateur female athletes with symptomatic FAI showed a lower alpha angle in young dance athletes compared to non-dancers (28), suggesting that certain young athletes who require extremes of motion to perform their sport may develop symptoms of impingement even in the absence of radiographically proven pincer or cam deformities.
FAI is being more commonly recognized and diagnosed in the pediatric and young adult population. Sink et al. (29) found the predominant clinical presentation in adolescents to be activity-related anterior groin pain. On exam, patients (85% female) exhibited decreased hip flexion and limited internal rotation, and all had a positive impingement test. Furthermore, most patients displayed mixed impingement (51%), followed by pincer impingement (43%) and cam impingement (6%). Nepple et al. (30) found distinct, sex-dependent disease patterns in patients with symptomatic FAI. Females had more profound symptomatology and milder morphologic abnormalities, while males had a higher activity level, larger morphologic abnormalities, more mixed type FAI morphologies and more extensive intra-articular disease. The correlation between FAI and osteoarthritis is gaining ground. Recently, Wyles et al. (31), showed after a 5-year follow-up that young athletes with cam deformities and limited ROM of the hip had progressive degenerative changes on MRI and radiographs consistent with early osteoarthritis, although the majority remained asymptomatic during the follow up period (see Figure 1 for an example of a prototypical cam morphology pre- and post-operatively in an adolescent male athlete).
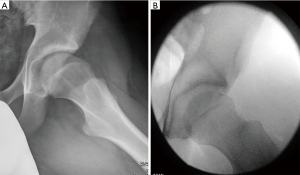
The results of arthroscopic treatment of FAI in the pediatric population have been encouraging. Byrd and colleagues found that 97% of patients reported improvement of their symptoms based on the Harris Hip Score post hip arthroscopy (32). This was reported on 116 hips with an average follow up of 38 months, with 94% of patients showing good or excellent results. Similarly, a retrospective study (33) of 60 pediatric and adolescent patients aged 16 years or younger who underwent hip arthroscopy showed improvement in the modified Harris Hip Score from a mean of 57 (95% CI, 51 to 62) to a mean of 91 (95% CI, 88 to 94) (P<0.001). However, 13% of patients required a second procedure for capsulolabral adhesions.
Studies have also shown positive and safe results using hip arthroscopy when addressing both the cam and acetabular rim deformities in skeletally immature patients (34,35). A small case series of eleven skeletally immature hips with open femoral head physes undergoing femoral osteochondroplasty showed high patient satisfaction and outcome scores. Furthermore, no evidence of bony regrowth at a minimum of two years post-operatively was found (36). Research focusing on the effectiveness of hip arthroscopy in the athletic adolescent population has shown promising results. Degen (37) followed young elite baseball players requiring hip arthroscopy for FAI and found a return to sport rate of 88% with 97% returning at or above their previous level of play. Meanwhile, a study of 51 professional football players reported that 87% were able to return to play as defined as competing in a pre-season or regular season professional football game after surgery (38). Overall, hip arthroscopy is a safe and effective tool for treating FAI and associated labral pathology in both the young athletic and non-athletic populations.
Acetabular labral tears
Labral tears in the hip can be a result of acute trauma, repetitive abutment secondary to FAI, or residual deformity from SCFE or other pediatric hip conditions. Labral tears can be successfully treated arthroscopically with repair, or when irreparable with debridement and/or labral reconstruction. It must be emphasized that particularly in young patients, the labrum should be preserved and repaired whenever possible (39,40).
Slipped capital femoral epiphysis (SCFE)
The deformity following a SCFE can lead to mild to severe cam-type deformity at the head neck junction from the resulting exposed proximal femoral metaphysis. There is some remodeling potential following in situ pinning of the SCFE, but this remodeling is limited especially in moderate to severe deformity (41,42). Repetitive trauma from the resulting FAI can lead to labral tears and chondral damage to the acetabulum and the femoral head. The traditional treatment for symptomatic residual deformity includes proximal femoral osteotomies (Southwick or Imhauser), surgical hip dislocation and the Modified Dunn osteotomy. More recently, hip arthroscopy has been used in the treatment of the cam lesion by performing a proximal femoral osteochondroplasty through a minimally invasive approach (43). This can be performed at the time of in-situ pinning, or staged at a later date if the patient becomes symptomatic (44). The argument for performing simultaneous pinning and osteochondroplasty is to avoid the potential cartilage and labral damage caused by the slip, although this has yet to be proven in long-term follow-up.
Ilizaliturri et al. reported on their results of arthroscopic management of FAI post SCFE (45) in eight patients (age 24–39 years) and at two-year minimum follow-up. The authors reported significant clinical improvements in post-operative Western Ontario McMasters Osteoarthritis (WOMAC) index with a mean increase of 9.6 points (4 to 14) (P=0.0001). Furthermore, none of the patients developed complications of avascular necrosis or fracture of the femoral neck. Since then, Chen et al., have reported larger series in younger patients and found that delayed treatment resulted in labral and cartilage damage, but that patients also experienced significant pain relief, improved internal rotation and improved radiographic alpha angles post arthroscopy (46). This study reported complications related to portal entry, capsular laxity and traction neuropraxias. In cases of larger deformity, some authors have suggested a variant of sub-capital realignment using arthroscopic techniques. This has demonstrated promising results in patient with deformities between 30 to 70 degrees, as measured by the epiphyseal-diaphyseal angle (47). However, in slips of greater magnitude, adequate posteromedial bone resection can be difficult to achieve arthroscopically due to posterior extension.
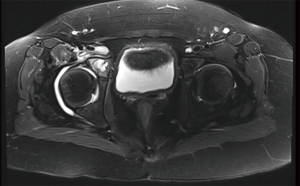
Finally, arthroscopy has been used in removal of hardware in preparation for proximal femoral osteoplasty (48). When considering arthroscopy for the treatment of SCFE, it is important to consider that arthroscopic osteoplasty is more technically and time-demanding than idiopathic FAI due to the severity of the deformity. It requires strong arthroscopic skills and experience in hip arthroscopy (see Figure 2 for an example of an arthroscopic SCFE deformity resection).
Septic arthritis
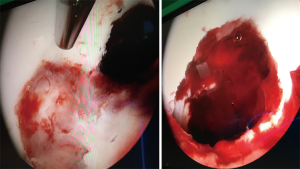
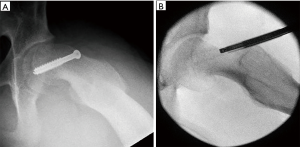
Septic arthritis of the pediatric hip is relatively common, and hip arthroscopy is increasingly being used for surgical treatment in pediatric centers where appropriate expertise and equipment exists. Currently, the gold standard for the treatment of septic arthritis consists of open anterior arthrotomy with associated irrigation and debridement. However, the potential benefits of hip arthroscopy over open arthrotomy are significant, and include more complete access to the joint, the ability to provide more aggressive irrigation and debridement, while still being significantly less invasive, with a lower risk of post-operative instability. In de Sa’s systematic review on arthroscopic management of the septic hip, five studies focused on pediatric patients, all with generally positive outcomes and no reported complications (49). El-Sayed compared irrigation and debridement by arthroscopy to open arthrotomy, and found patients were discharged significantly faster with arthroscopic management (3.8 days to discharge vs. 6.4, respectively) (50). Septic arthritis can be secondary to extension of adjacent osteomyelitis of the acetabulum or femoral head and neck. This is a particularly common scenario in neonates who have transphyseal vessels that allow for spread into the joint. These can provide significant challenges for surgical management via open techniques as adequate access and exposure can require very invasive approaches. Alternatively, arthroscopy can again provide better access to these hard to reach places, with excellent visualization and options for extended tools that can be used to assist with necessary debridement (see Figures 3,4 for an example of a septic hip and adjacent acetabular osteomyelitis in an 8-year-old, treated successfully with arthroscopic irrigation and debridement). As previously stated, patients with septic arthritis are typically younger, and important considerations for a young child include the question of equipment availability and size, and experience of the surgeon. A traditional hip distractor may not be necessary, depending on the child and the degree of effusion. Furthermore, certain infections may not require access to the central compartment, negating the need for distraction. Using portals that safely optimize access to the peripheral compartment is a technical pearl to successful outcomes.
Developmental dysplasia of the hip (DDH)
The use of hip arthroscopy for the treatment of DDH has evolved considerably over the last decade. Initially used to treat labral tears in the presence of acetabular dysplasia, recent studies have shown less favorable results when the indications for arthroscopy alone are stretched and it is used for severe dysplasia; therefore, pelvic osteotomies remain the gold standard of treatment for dysplasia (21). Recent publications have shown that periacetabular osteotomies in combination with hip arthroscopy can respectfully address the bony deformity and labral pathology associated with dysplasia in a safe and effective manner (21). A study of patients with borderline dysplasia [lateral center edge angle (LCEA) of 20–25 degrees] who underwent arthroscopic capsular plication showed favorable results in all clinical scores at 2-year follow-up (51). This study however included a heterogeneous mixture of concomitant procedures that included femoral osteoplasty, chondroplasty, and ligamentum teres debridement. Finally, arthroscopy in the patient with DDH can also be used to aid in the early stages with hip reduction. A case series by Xu and colleagues included 44 hips with DDH in 35 patients for which arthroscopic-assisted reduction of the hip was performed after failed closed reduction (52). Arthroscopic treatment was successfully performed in 40 hips, while the remaining 4 hips required open reduction. The arthroscope was helpful in visualizing and removing obstacles to reduction, such as resection of pulvinar, ligamentum teres excision and transverse acetabular ligament release.
Complications
Although hip arthroscopy continues to be an advantageous tool in the diagnosis and treatment of various hip pathologies, associated risks do exist and need to be reviewed with patients and their families. Post-operative complications have predominantly been documented in the adult population, however a more recent case series by Nwachukwu and colleagues has been published on pediatric patients (53). This study reported on 218 pediatric arthroscopies for the treatment of labral tears, loose bodies, FAI and SCFE. The complication rate was 1.8%, with transient pudendal nerve neuropraxia being the predominant complication, followed by instrument breakage and suture abscess. There was no documentation of growth arrest, physeal injury, or osteonecrosis during this series. Similarly, in a systematic review of eight studies looking at hip arthroscopy for adolescent patients with FAI, there were no documented complications of physeal arrest, infection, AVN or instrument breakage (54). Of the 435 hip scopes, there were two cases of transient perineal nerve neuropraxia, one case of heterotopic ossification (HO) not requiring further intervention, and 13 cases of capsular adhesions that responded well to revision surgery. This resulted in a 3.6% rate of minor complications from hip arthroscopy for adolescent patients with FAI. Furthermore, this review concluded that complications in the adolescent population seem to differ from those of adults as there were no documented cases of fluid extravasation, iatrogenic injury secondary to portal placement, deep vein thrombosis (DVT), or vascular injury. Transient pudendal nerve neuropraxia is best avoided by limiting traction time and force, and having a well-padded perineal post (55). Should a neuropraxia develop, the majority appear to resolve spontaneously within 3 months (56). In the prevention of HO, Beckmann (57) demonstrated in a randomized trial a significant reduction in post-operative HO when treated prophylactically with Naproxen twice daily for 3 weeks (4%) compared to placebo (44%). However, the difference in clinical outcomes with and without prophylaxis has not been adequately studied, and the role of post-operative prophylactic NSAIDs is still debated given the potential benefits versus risk profile.
Conclusions
Hip arthroscopy is an excellent technique for visualizing and treating many pediatric and adolescent hip disorders, either directly or as an adjunct. For many conditions it provides unparalleled surgical access and allows for safe and effective procedures that can be used to address various intra-articular, as well as, extra-articular pathologies. It is important to clearly understand not only the benefits of arthroscopy, but also the technical limitations. Acquiring the necessary skills takes practice, sufficient patient volumes, and the necessary equipment and support. In the right circumstances and setting, it is an extremely useful tool that can be used to improve care in a growing number of pediatric pathologies.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Olufemi R. Ayeni and Ryan P. Coughlin) for the series “Future Perspectives in Hip Preservation and Arthroscopy” published in Annals of Joint. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2018.06.01). The series “Future Perspectives in Hip Preservation and Arthroscopy” was commissioned by the editorial office without any funding or sponsorship. SC reports grants from Zimmer Biomet, grants from ConMed Linvatec, outside the submitted work. KS reports grants from Zimmer BioMet, outside the submitted work. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Bozic KJ, Chan V, Valone FH 3rd, et al. Trends in hip arthroscopy utilization in the United Staes. J Arthroplasty 2013;28:140-3. [Crossref] [PubMed]
- Gross RH. Arthroscopy in hip disorders in children. Orthop Rev 1977;6:43-9.
- Matsuda DK, Carlisle JC, Arthurs SC, et al. Comparative systematic review of the open dislocation, mini-open, and arthroscopic surgeries for femoroacetabular impingement. Arthroscopy 2011;27:252-69. [Crossref] [PubMed]
- Botser IM, Smith TW Jr, Nasser R, et al. Open surgical dislocation versus arthroscopy for femoroacetabular impingement: a comparison of clinical outcomes. Arthroscopy 2011;27:270-8. [Crossref] [PubMed]
- Byrd JW. Femoroacetabular impingement in athletes: Part 1. Cause and assessment. Sports Health 2010;2:321-33. [Crossref] [PubMed]
- Byrd JW. Evaluation of the Hip: History and Physical Examination. N Am J Sports Phys Ther 2007;2:231-40. [PubMed]
- Matava MJ, Patton CM, Luhmann S, et al. Knee pain as the initial symptom of slipped capital femoral epiphysis: an analysis of initial presentation and treatment. J Pediatr Orthop 1999;19:455-60. [Crossref] [PubMed]
- Yen YM, Lewis CL, Kim YJ. Understanding and Treating the Snapping Hip. Sports Med Arthrosc Rev 2015;23:194-9. [Crossref] [PubMed]
- Tijssen M, van Cingel R, Willemsen L, et al. Diagnostics of femoroacetabular impingement and labral pathology of the hip: a systematic review of the accuracy and validity of physical tests. Arthroscopy 2012;28:860-71. [Crossref] [PubMed]
- Reiman MP, Goode AP, Cook CE, et al. Diagnostic accuracy of clinical tests for the diagnosis of hip femoroacetabular impingement/labral tear: a systematic review with meta-analysis. Br J Sports Med 2015;49:811. [Crossref] [PubMed]
- Haldane CE, Ekhtiari S, de Sa D, et al. Preoperative physical examination and imaging of femoroacetabular impingement prior to hip arthroscopy—a systematic review. J Hip Preserv Surg 2017;4:201-13. [Crossref] [PubMed]
- DeAngelis N, Busconi B. Assessment and differential diagnosis of the painful hip. Clin Orthop Relat Res 2003;11-8. [Crossref] [PubMed]
- Clohisy JC, Carlisle JC, Beaulé PE, et al. A Systematic Approach to the Plain Radiographic Evaluation of the Young Adult Hip. J Bone Joint Surg Am 2008;90:47-66. [Crossref] [PubMed]
- Wylie JD, Jenkins PA, Beckmann JT, et al. Computed tomography scans in patients with young adult hip pain carry a lifetime risk of malignancy. Arthroscopy 2018;34:155-63.e3. [Crossref] [PubMed]
- Geijer M, Rundgren G, Weber L, et al. Effective dose in low-dose CT compared with radiography for templating of total hip arthroplasty. Acta Radiol 2017;58:1276-82. [Crossref] [PubMed]
- Linda DD, Naraghi A, Murnaghan L, et al. Accuracy of non-arthrographic 3T MR imaging in evaluation of intra-articular pathology of the hip in femoroacetabular impingement. Skeletal Radiol 2017;46:299-308. [Crossref] [PubMed]
- Kocher MS, Kim YJ, Millis MB, et al. Hip arthroscopy in children and adolescents. J Pediatr Orthop 2005;25:680-6. [Crossref] [PubMed]
- Mandell JC, Marshall RA, Banffy MB, et al. Arthroscopy after traumatic hip dislocation: a systematic review of intra-articular findings, correlation with magnetic resonance imaging and computed tomography, treatments, and outcomes. Arthroscopy 2018;34:917-27. [Crossref] [PubMed]
- Hwang JM, Hwang DS. Hip arthroscopy for incarcerated acetabular labrum following reduction of traumatic hip dislocation: three case reports. Hip Pelvis 2016;28:164-8. [Crossref] [PubMed]
- Domb BG, Chaharbakhshi EO, Perets I, et al. Hip arthroscopic surgery with labral preservation and capsular plication in patients with borderline hip dysplasia: minimum 5-Year patient-reported outcomes. Am J Sports Med 2018;46:305-13. [Crossref] [PubMed]
- Ricciardi BF, Fields KG, Wentzel C, et al. Early functional outcomes of periacetabular osteotomy after failed hip arthroscopic surgery for symptomatic acetabular dysplasia. Am J Sports Med 2017;45:2460-7. [Crossref] [PubMed]
- Ekhtiari S, de Sa D, Haldane CE, et al. Hip arthroscopic capsulotomy techniques and capsular management strategies: a systematic review. Knee Surg Sports Traumatol Arthrosc 2017;25:9-23. [Crossref] [PubMed]
- Siebenrock KA, Behning A, Mamisch TC, et al. Growth plate alteration precedes cam-type deformity in elite basketball players. Clin Orthop Relat Res 2013;471:1084-91. [Crossref] [PubMed]
- Kapron AL, Anderson AE, Aoki SK, et al. Radiographic prevalence of femoroacetabular impingement in collegiate football players: AAOS Exhibit Selection. J Bone Joint Surg Am 2011;93:e111(1-10).
- Siebenrock KA, Kaschka I, Frauchiger L. Prevalence of cam-type deformity and hip pain in elite ice hockey players before and after the end of growth. Am J Sports Med 2013;41:2308-13. [Crossref] [PubMed]
- Agricola R, Heijboer MP, Abida Z, et al. A cam deformity is gradually acquired during skeletal maturation in adolescent and Young male soccer players: A Prospective study with mínimum 2-year follow up. Am J Sports Med 2014;42:798-806. [Crossref] [PubMed]
- Carsen S, Moroz PJ, Rakhra K, et al. The otto aufranc award. On the etiology of the cam deformity: A cross-sectional pediatric MRI study. Clin Orthop Relat Res 2014;472:430-6. [Crossref] [PubMed]
- Fraser JL, Sugimoto D, Beng Y, et al. Bony morphology of femoroacetabular impingement in young female dancers and single-sport athletes. Orthop J Sports Med 2017;5:2325967117723108 [Crossref] [PubMed]
- Sink EL, Gralla J, Ryba A, et al. Clinical presentation of femoroacetabular impingement in adolescents. J Pediatr Orthop 2008;28:806-11. [Crossref] [PubMed]
- Nepple JJ, Riggs CN, Ross JR, et al. Clinical presentation and disease characteristics of femoroacetabular impingement are sex-dependent. J Bone Joint Surg Am 2014;96:1683-9. [Crossref] [PubMed]
- Wyles CC, Norambuena GA, Howe BM, et al. Cam deformities and limited hip range of motion are associated with early osteoarthritic changes in adolescent athletes: A prospective matched cohort study. Am J Sports Med 2017;45:3036-43. [Crossref] [PubMed]
- Byrd JW, Jones KS, Gwathmey FW, et al. Femoroacetabular Impingement in Adolescent Athletes: Outcomes of Arthroscopic Management. Am J Sports Med 2016;44:2106-11. [Crossref] [PubMed]
- Philippon MJ, Ejnisman L, Ellis HB, et al. Outcomes 2 to 5 years following hip arthroscopy for femoroacetabular impingement in the patient aged 11 to 16 years. Arthroscopy 2012;28:1255-61. [Crossref] [PubMed]
- Papalia R, Del Buono A, Franceschi F, et al. Femoroacetabular impingement syndrome management: arthroscopy or open surgery. Int Orthop 2012;36:903-14. [Crossref] [PubMed]
- Byrd JW, Jones KS. Arthroscopic femoroplasty in the management of Cam-type femorocetabular impingement. Clin Orthop Relat Res 2009;467:739-46. [Crossref] [PubMed]
- Perets I, Gupta A, Chaharbakhshi EO, et al. Does bony regrowth occur after arthroscopic femoroplasty in a group of young adolescents? Arthroscopy 2017;33:988-95. [Crossref] [PubMed]
- Degen RM, Fields KG, Wentzel CA, et al. Return-to-play rates following arthroscopic treatment of femoroacetabular impingement in competitive baseball players. Phys Sportsmed 2016;44:385-90. [Crossref] [PubMed]
- Menge TJ, Bhatia S. Femoroacetabular impingement in professional football players return to play and predictors of career length after hip arthroscopy. Am J Sports Med 2017;45:1740-4. [Crossref] [PubMed]
- Clohisy JC, Baca G, Beaulé PE. Descriptive epidemiology of femoroacetabular impingement: a North American cohort of patients undergoing surgery. Am J Sports Med 2013;41:1348-56. [Crossref] [PubMed]
- Nawabi DH, Degen RM, Fields KG, et al. Outcomes after arthroscopic treatment of femoroacetabular impingement for patients with borderline hip dysplasia. Am J Sports Med 2016;44:1017-23. [Crossref] [PubMed]
- Dawes B, Jaremko JL, Balakumar J. Radiographic assessment of bone remodelling in slipped upper femoral epiphyses using Klein’s line and the alpha angle of femoral-acetabular impingement: a retrospective review. J Pediatr Orthop 2011;31:153-8. [Crossref] [PubMed]
- Akiyama M, Nakashima Y, Kitano T, et al. Remodelling of femoral head-neck junction in slipped capital femoral epiphysis: a multi-centre study. Int Orthop 2013;37:2331-6. [Crossref] [PubMed]
- Oduwole K, de Sa D, Kay J, et al. Surgical treatment of femoroacetabular impingement following slipped capital femoral epiphysis: A systematic review. Bone Joint Res 2017;6:472-80. [Crossref] [PubMed]
- Accadbled F, May O, Thevenin-Lemoine C, et al. Slipped capital femoral epiphysis management and the arthroscope. J Child Orthop 2017;11:128-30. [Crossref] [PubMed]
- Ilizaliturri VM Jr, Nossa-Barrera JM, Acosta-Rodriguez E, et al. Arthroscopic treatment of femoroacetabular impingement secondary to paediatric hip disorders. J Bone Joint Surg Br 2007;89:1025-30. [Crossref] [PubMed]
- Chen A, Youderian A, Watkins S, et al. Arthroscopic Femoral Neck Osteoplasty in Slipped Capital Femoral Epiphysis. Arthroscopy 2014;30:1229-34. [Crossref] [PubMed]
- Dutra Roos B, Camargo de Assis M, Roos MC, et al. Arthroscopic subcapital realignment in chronic and stable slipped capital femoral epiphysis. Arthrosc Tech 2017;6:e667-72. [Crossref] [PubMed]
- Howse EA, Wooster BM, Mannava S, et al. Arthroscopic treatment of slipped capital femoral epiphysis screw impingement and concomitant hip pathology. Arthrosc Tech 2014;3:e515-7. [Crossref] [PubMed]
- de Sa D, Cargnelli S, Catapano M, et al. Efficacy of hip arthroscopy for the management of septic arthritis: A systematic review. Arthroscopy 2015;31:1358-70. [Crossref] [PubMed]
- El-Sayed AM. Treatment of early septic arthritis of the hip in children: comparison of results of open arthrotomy versus arthroscopic drainage. J Child Orthop 2008;2:229-37. [Crossref] [PubMed]
- Domb BG, Stake CE, Lindner D, et al. Arthroscopic Capsular Plication and Labral Preservation in Hip Dysplasia. Am J Sports Med 2013;41:2591-8. [Crossref] [PubMed]
- Xu HF, Yan YB, Xu C. Effects of arthroscopic-assisted surgery on irreducible developmental dislocation of hip by mid-term follow-up: An observational study. Medicine (Baltimore) 2016;95:e4601 [Crossref] [PubMed]
- Nwachukwu BU, McFeely ED, Nasreddine AY, et al. Complications of hip arthroscopy in children and adolescents. J Pediatr Orthop 2011;31:227-31. [Crossref] [PubMed]
- de Sa D, Cargnelli S, Catapano M, et al. Femoroacetabular impingement in skeletally immature patients: a systematic review examining indications, outcomes, and complications of open and arthroscopic treatment. Arthroscopy 2015;31:378-84. [Crossref] [PubMed]
- Frandsen L, Lund B, Gronbech N, et al. Traction-related problems after hip arthroscopy. J Hip Preserv Surg 2017;4:54-9. [PubMed]
- Habib A, Haldane CE, Ekhtiari S, et al. Pudendal nerve injury is a relatively common but transient complication of hip arthroscopy. Knee Surg Sports Traumatol Arthrosc 2018;26:969-75. [Crossref] [PubMed]
- Beckmann JT, Wylie JD, Potter MQ, et al. Effect of naproxen prophylaxis on heterotopic ossification following hip arthroscopy: A double-blind randomized placebo-controlled rial. J Bone Joint Surg AM 2015;97:2032-7. [Crossref] [PubMed]
Cite this article as: Carsen S, Tourn D, Marien M, Smit K. Arthroscopy of the pediatric hip. Ann Joint 2018;3:58.


