
Is obesity a risk factor for extended length of stay and readmission after total hip arthroplasty?
Introduction
Total hip arthroplasties (THA) are common, effective surgical procedures that increase patient and societal productivity, even resulting in a financial benefit for society. This benefit to society will continue to grow as the number of THAs performed each year increases as the baby boomer cohort ages and its members wish to maintain active lifestyles. This growth is demonstrated by joint registry data which has shown 2 million Americans are expected to receive THA or total knee arthroplasty (TKA) in 2020 and an increase in THA revision rates since 2008 (1). Although these are effective surgeries, they are sometimes associated with significant and costly complications that can result in an increased hospital length of stay (LOS) and increased readmission rates (RR) (2). Readmissions following THAs account for 12% of reimbursements spent on post-arthroplasty hospital readmissions (3). Due to the increasing demand for THAs and their associated complications, hospitals and health insurance entities, especially the Centre for Medicare and Medicaid services, place a great deal of emphasis on identifying and minimizing modifiable risk factors (4).
In a movement towards cost containment, voluntary programs such as the Medicare Bundled Payment for Care Improvement initiative are offering single, prospectively determined bundled payments to hospitals that cover all of the services offered during a single episode of care (the length of an entire inpatient stay). This program seeks to discourage unnecessary services and billing associated with the fee-for-service model and instead incentivizes coordinated and efficient care (5). More recently, the Comprehensive Care for Joint Replacement (CJR) model, effective April 1st 2016, offers bundle payments for hip and knee arthroplasties to cover care through 90 days after hospital discharge (6).
Considering these new methods of reimbursement centered on coordinated care and the burden of THA associated complications on individual patients and on society, it is imperative to accurately identify high risk patients and successfully implement clinical interventions to address modifiable risk factors before performing the operations (7). Commonly studied risk factors for THA complications are diabetes mellitus (DM), hypertension (HTN), coronary artery disease (CAD), congestive heart failure (CHF), peripheral vascular disease (PVD), chronic obstructive pulmonary disease (COPD), renal disease, mental illness, asthma, and obesity (7). Obesity in particular has received attention as a prevalent and modifiable risk factor.
While obesity as an epidemic is widely accepted, the exact role of obesity in THA complications in particular has been a subject of debate. Obesity is a worldwide epidemic, affecting as much as 70% of males and 56% of females (8). In the USA, 66% of adults are classified as obese with a 50% annual increase in the prevalence of body mass index (BMI) >40 kg/m2 (9). This increase in obesity is of particular importance to THAs and their associated increase in LOS and RR as there is a strong association between obesity and joint disease of the lower limbs (10).
There have been conflicting results on the association between obesity and complications following THA. Bradley et al. (11) followed 589 patients undergoing lower limb arthroplasty and found that for every point increase in BMI the LOS increased by a factor of 2.9% (P<0.0001). Conversely, Deakin et al. (12) found no increase in LOS for obese patients compared on non-obese patients. Ali et al. (13) found that obese patients had a 1.14 higher risk (P<0.0001) for readmission while Shaparin et al. (14) found RR were not significantly different for patients with a BMI of 30–34.9 kg/m2 compared to patients with a BMI of 25–25.9 kg/m2 (P=0.9).
The purpose of this study was to determine if obesity as an independent risk factor was associated with an increased LOS and RR. The second purpose of this study was to evaluate obesity as a risk factor when compared to other comorbidities.
Methods
This case-control study was approved by our institutional review board (IRB approval 849-16-EP). Data was gathered from the electronic medical records of all patients who were admitted following a primary THA at our University Medical Centre from November 2009 to November 2016 (7 years). Surgeries were performed by three separate physicians within the institution with a minimum of 1-year follow-up. Our search yielded 1,447 THA surgeries during this time period, of which 14 encounters were excluded due to incomplete chart information (12) or death during hospitalization (2). The 1,433 surgeries were performed on 1,265 patients. Readmission was defined as any unplanned, any-cause hospitalization up to 90 days after discharge from initial post-THA hospitalization to our facilities. Patients and data from other institutions were not included in this study. All patients who were included in analysis had BMI and comorbidity data available at the time of surgery. BMI was calculated using the formula weight in kilograms divided by the square of the height in meters (kg/m2). BMI ranges were defined using the National Institutes of Health Patients ranges as follows: underweight (BMI ≤18.5 kg/m2), normal weight (BMI =18.6–24.9 kg/m2), overweight (BMI =25.0–29.9 kg/m2), obese class I (BMI =30.0–34.9 kg/m2), obese class II (BMI =35.0–39.9 kg/m2), and obese class III (BMI ≥40 kg/m2) (15).
Variables of interest were collected through chart review including age, gender, race, BMI, comorbidities (HTN, asthma, DM, renal disease, mental illness, COPD, PVD, CAD, CHF), LOS, readmission, discharge location and cause of readmission. Specifically, BMI and comorbidity data were collected from the vitals and past medical history sections of the pre-surgical history and physical.
Statistical analysis
Counts and percentages were used to describe categorical data such as pre-surgery demographics, comorbidities, and RR. Means and standard deviations were used to describe continuous data such as age, LOS, and BMI. BMI was also evaluated as a categorical variable when determining associations with potential confounders as the various BMI categories: underweight, normal weight, overweight, obese class I, obese class II, obese class III. Analysis of variables was used to examine the association of age, gender, race, LOS, RR, HTN, asthma, DM, renal disease, mental illness, COPD, PVD, CAD, CHF with each of the six BMI categories. Pairwise comparisons were adjusted using Tukey’s method. For each BMI category and for the potential confounders, we calculated the odds ratio of increased risk of readmission and increased LOS (≥5 days). A cut-off of ≥5 days for LOS was considered an extended stay because it was approximately one standard deviation above the mean. Odds ratios were calculated for the BMI categories with the overweight category as a reference.
A multivariable logistic regression model was fit that included age and all conditions considered in the univariate analysis. From this complete model, a reduced model was developed that included the most predictive variables of readmission as well as BMI status (either ≥40 or ≥50 kg/m2 cut point) using backward elimination until the remaining variables were significant at the P<0.05 level. This backward elimination excluded the variables that were not statistically significant. BMI was forced into the model as it was the variable of interest. The following variables were included in the final multivariate analysis: age, HTN, CAD, CHF, PVD, COPD, asthma, renal disease, and mental illness. This analysis was used to examine BMI ≥40 or ≥50 kg/m2 as independent risk factors for readmission by adjusting for the possible confounding demographic and comorbidity variables. A multivariate analysis was not performed for LOS because BMI was not significantly associated with it in univariate analysis.
Because our study spanned 7 years, we examined the association between the main variables of interest, LOS, RR, and BMI, with each year of the study to account for potential changes in surgery methods or post-surgical medical care that may have occurred over the period of the study.
All analyses were performed using SAS 9.4 and a P value <0.05 was considered statistically significant.
Results
Patients
Of the 1,433 surgeries, 622 (43.4%) were male and 811 (56.6%) were female; 1,291 (90.1%) white/non-Hispanic, 104 (7.3%) black, 15 (1.0%) Hispanic, 10 (0.7%) Asian, and 13 (0.9%) deferred their race. Age for all encounters was normally distributed with a mean of 60.7±13.8 years and a median of 61.0 (range, 12.0–94.0) years. BMI for all encounters was skewed to the right with a mean of 30.0±6.7 kg/m2 and a median of 28.8 (range, 13.9–68.8) kg/m2. Distribution between the BMI categories was as follows: underweight 1.3% (n=19), normal weight 20.8% (n=298), overweight 33.8% (n=484), obese class I 22.5% (n=323), obese class II 13.2% (n=189), and obese class III 8.4% (n=120). LOS for all encounters was skewed to the right with a mean of 3.3±1.9 days and a median of 3.0 (range, 1.0–28.0) days. The RR for all encounters was 6.6% (n=94). The frequencies for all encounters of the various comorbidities are as follows: HTN 55.7% (n=798), asthma 10.1% (n=145), DM 15.0% (n=215), renal disease 10.5% (n=150), mental illness 22.3% (n=320), COPD 5.3% (n=76), PVD 5.7% (n=81), CAD 11.3% (n=162), and CHF 3.9% (n=56).
BMI category associations
Age was significantly associated with BMI categories (P<0.0001), the mean age decreasing with increasing BMI (Table 1). Gender was also significantly associated with BMI categories (P<0.0001), with the distribution of a larger percentage of males in the middle categories, overweight and obese class I, and a larger percentage of females in the categories underweight, normal weight, obese class II and obese class III (Table 1). In the category of race, increasing BMI was associated with an increased percentage of blacks (P=0.015) and decreasing BMI was associated with an increased percentage of whites (P=0.006) (Table 1). The other races were not significantly associated. The following comorbidities were significantly associated with BMI categories: HTN (P<0.0001), DM (P<0.0001), mental illness (P=0.006), COPD (P<0.0001), and CAD (P=0.023). The following comorbidities were not significantly associated with BMI categories: asthma (P=0.253), renal disease (P=0.210), PVD (P=0.838), and CHF (P=0.149) (Table 1). Although LOS was significantly associated BMI (P=0.046), the lowest mean LOS of 3.1±1.3 days was found in the obese class I category while the highest mean LOS of 3.5±2.5 days was found in the normal weight category (Table 1). RR were not significantly associated with BMI categories (P=0.308), but there was a large difference between the lowest RR of 5.3% for obese class II and 11.7% for obese class III (Table 1).
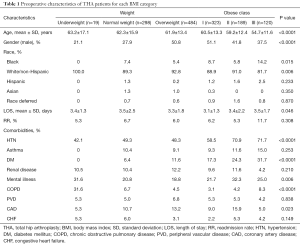
Full table
Predictors of readmission
By univariate analysis, the following variables were significantly associated with an increased odds of readmission: black race (P=0.001), obese class III (P=0.034), HTN (P=0.0002), asthma (P=0.010), renal disease (P<0.0001), mental illness (P=0.001), COPD (P<0.0001), PVD (P<0.0001), CAD (P<0.0001), and CHF (P=0.0002). Those not significantly associated with increased risk of readmission were gender (P=0.213), underweight (P=0.895), normal weight (P=0.687), overweight (P=1.000), obese class I (P=0.907), obese class II (P=0.727), and DM (P=0.146) (Table 2).
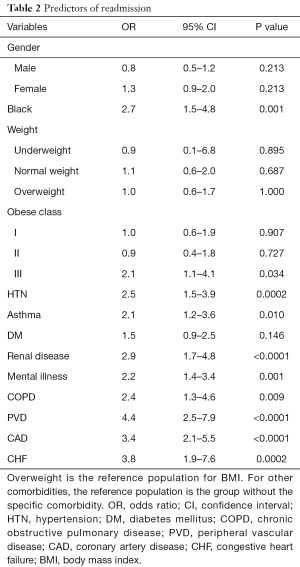
Full table
By multivariate analysis with adjustment for confounders using a cutoff of BMI 40 kg/m2, there was no significant increase in the independent odds of readmission for BMI ≥40 kg/m2 compared to BMI <40 kg/m2 (P=0.621) (Table 3). With a cutoff of BMI 50 kg/m2, there was a significant increase in the independent odds of readmission for BMI ≥50 kg/m2 compared to BMI <50 kg/m2 (P=0.015), but the sample size for BMI ≥50 kg/m2 was only n=11 (Table 4).
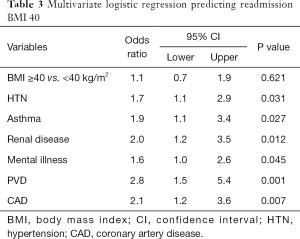
Full table
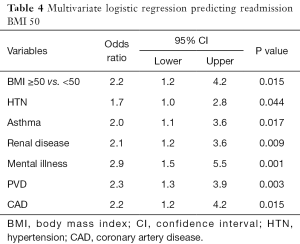
Full table
Predictors of increased LOS
By univariate analysis, the following variables were significantly associated with an increased odds of LOS ≥5 days: asthma (P=0.011), DM (P=0.003), renal disease (P=0.007), mental illness (P=0.021), COPD (P=0.0004), PVD (P=0.003), CAD (0.001), and CHF (P<0.0001). Those not significantly associated with increased risk of extended LOS were gender (P=0.282), black race (P=0.369), underweight (P=0.246), normal weight (P=0.649), overweight (P=1.000), obese class II (P=0.610), obese class III (P=0.877), and HTN (P=0.098). Obese class I actually had a reduced risk of extended LOS compared to overweight (P=0.005) (Table 5).
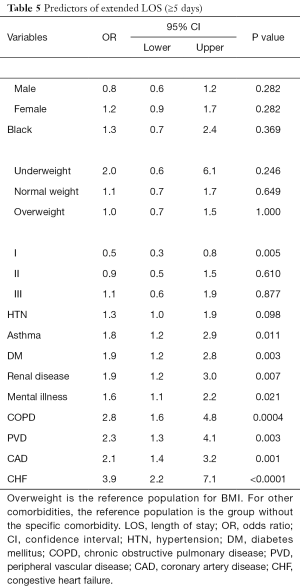
Full table
Because BMI was not associated with increased odds of extended LOS at any level, no multivariate analysis was performed.
LOS, RR, and BMI over time of study
There was a significant difference in the LOS over the course of the study. The mean LOS fluctuated over time with the greatest value of 3.6±1.4 days in 2011 to least value of 3.0±3.0 days in 2016 (P=0.024) (Table 6). The RR (P=0.220) and the BMI (P=0.541) did not change significantly over the course of the study (Table 6).

Full table
Discussion
Obesity is a serious epidemic that has profound impacts on the healthcare system. Thompson et al. (16) demonstrated that lifetime medical costs increased by 20% for mild obesity, 50% for moderate obesity and 200% for severe obesity. This rise in obesity has coincided with a rise in the demand for THAs due to the increased likelihood of arthritis in the obese compared to the general population. The number of obese patients presenting for joint arthroplasties has been increasing over time (17). These facts, in combination with continued efforts to minimize healthcare costs, make it imperative to determine the relationship between obesity and complications following joint arthroplasties. The current research has provided conflicting results on this relationship. Studies have shown little to no significant difference in the outcomes between obese and non-obese patients (12,14) while others have shown significant difference (11,13,18). More research is needed to define the specific impacts of obesity on patient outcomes.
This study focused on obesity’s impact on the outcomes of LOS and RR and found no significant relationship between obesity and LOS nor RR below BMI of 50 following a primary THA. There was an increased odds of readmission with BMI ≥50 (P=0.015), although the sample size for this group was small (n=11) so it is difficult to make a firm conclusion for this group. However, several comorbidities including asthma, CAD, CHF, PVD, COPD, and mental illness were all associated with both increased odds of extended LOS and readmission. HTN was associated with increased odds of readmission only and diabetes was associated with increased odds of extended LOS only. When compared to the other comorbidities in this study, obesity had no significant impact on LOS nor readmission below BMI of 50, even when considered as an independent factor. This suggests that efforts to minimize cost and complications for THAs should focus on optimizing the comorbidities associated with extended LOS and readmission prior to surgery, rather than on obesity.
Although obesity is associated with other independent risk factors like malnutrition (19) and physical weakness (20), the findings of this study question whether or not obesity itself should be considered a stand-alone risk factor for THA. Our study is not alone in asserting that LOS and RR are not increased in patients with obesity (unless BMI ≥50) (12,14). However, largely elevated BMIs may need to be considered a stand-alone risk factor as Arsoy et al. found a higher complication risk for super obese patients (BMI ≥50) compared to non-obese patients (BMI <25) (21).
Conclusions
This study demonstrated that the minimal effect BMI has on LOS and RR that is not consistent with the strong bias and association of obesity with perioperative complications, asserting that obesity (BMI, 30–40 kg/m2) is not an independent risk factor for increased LOS and RR following THA. Patients with a BMI ≥50 kg/m2 are an exception as this population trended toward an odds of readmission after adjusting for confounders, although the population size was small for this group and more research is needed at this BMI level.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2018.08.03). KG serves as an unpaid editorial board member of Annals of Joint from May 2018 to Apr 2020. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Informed consent was taken from all subjects. This study was approved by our Institutional Review Board (No. 849-16-EP).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kurtz SM, Ong KL, Lau E, et al. Impact of the economic downturn on total joint replacement demand in the United States: updated projections to 2021. J Bone Joint Surg Am 2014;96:624-30. [Crossref] [PubMed]
- Mednick RE, Alvi HM, Krishnan V, et al. Factors Affecting Readmission Rates Following Primary Total Hip Arthroplasty. J Bone Joint Surg Am 2014;96:1201-9. [Crossref] [PubMed]
- Bosco JA 3rd, Karkenny AJ, Hutzler LH, et al. Cost burden of 30-day readmissions following Medicare total hip and knee arthroplasty. J Arthroplasty 2014;29:903-5. [Crossref] [PubMed]
- Services CfMaM. Bundled payments for care improvement (BPCI) initiative: general information. Accessed 12/20 2017. Available online: http://innovation.cms.gov/initiatives/bundled-payments/
- Services CfMaM. Readmission reduction program. Accessed 12/20 2017. Available online: http://www.cms.gov/Medicare/Meicare-Fee-for-Service-Payment/AcuteInpatientPPS/Readmissions-Reduction-Program.html
- Services CfMaM. Comprehensive care for joint replacement model. Accessed 11/11 2015. Available online: https://innovation.cms.gov/initiatives/cjr
- Yu S, Garvin KL, Healy WL, et al. Preventing Hospital Readmissions and Limiting the Complications Associated With Total Joint Arthroplasty. J Am Acad Orthop Surg 2015;23:e60-71. [Crossref] [PubMed]
- Australian Orthopaedic Association National Joint Replacement Registry. Hip and Knee Replacement. Available online: http://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/4364.0.55.001~2014-15~Main%20Features~Overweight%20and%20obesity~22
- Smith KB, Smith MS. Obesity Statistics. Prim Care 2016;43:121-35. ix. [Crossref] [PubMed]
- Tukker A, Visscher TL, Picavet HS. Overweight and health problems of the lower extremities: osteoarthritis, pain and disability. Public Health Nutr 2009;12:359-68. [PubMed]
- Bradley BM, Griffiths SN, Stewart KJ, et al. The effect of obesity and increasing age on operative time and length of stay in primary hip and knee arthroplasty. J Arthroplasty 2014;29:1906-10. [Crossref] [PubMed]
- Deakin AH, Iyayi-Igbinovia A, Love GJ. A comparison of outcomes in morbidly obese, obese and non-obese patients undergoing primary total knee and total hip arthroplasty. Surgeon 2018;16:40-5. [Crossref] [PubMed]
- Ali AM, Loeffler MD, Aylin P, et al. Factors Associated With 30-Day Readmission After Primary Total Hip Arthroplasty: Analysis of 514455 Procedures in the UK National Health Service. JAMA Surg 2017;152:e173949 [Crossref] [PubMed]
- Shaparin N, Widyn J, Nair S, et al. Does the obesity paradox apply to early postoperative complications after hip surgery? A retrospective chart review. J Clin Anesth 2016;32:84-91. [Crossref] [PubMed]
- National Institutes of H. nhlbi support. Available online: http://nhlbisupport.com/bmi/bminojs.htm
- Thompson D, Edelsberg J, Colditz GA, et al. Lifetime health and economic consequences of obesity. Arch Intern Med 1999;159:2177-83. [Crossref] [PubMed]
- Fehring TK, Odum SM, Griffin WL, et al. The obesity epidemic: its effect on total joint arthroplasty. J Arthroplasty 2007;22:71-6. [Crossref] [PubMed]
- Wagner ER, Kamath AF, Fruth KM, et al. Effect of Body Mass Index on Complications and Reoperations After Total Hip Arthroplasty. J Bone Joint Surg Am 2016;98:169-79. [Crossref] [PubMed]
- Fu MC, D'Ambrosia C, McLawhorn AS, et al. Malnutrition Increases With Obesity and Is a Stronger Independent Risk Factor for Postoperative Complications: A Propensity-Adjusted Analysis of Total Hip Arthroplasty Patients. J Arthroplasty 2016;31:2415-21. [Crossref] [PubMed]
- Oosting E, Hoogeboom TJ, Dronkers JJ, et al. The Influence of Muscle Weakness on the Association Between Obesity and Inpatient Recovery From Total Hip Arthroplasty. J Arthroplasty 2017;32:1918-22. [Crossref] [PubMed]
- Arsoy D, Woodcock JA, Lewallen DG, et al. Outcomes and complications following total hip arthroplasty in the super-obese patient, BMI > 50. J Arthroplasty 2014;29:1899-905. [Crossref] [PubMed]
Cite this article as: Richard B, Reilly A, Lyden E, Garvin K. Is obesity a risk factor for extended length of stay and readmission after total hip arthroplasty? Ann Joint 2018;3:73.


