
Periprosthetic joint infection in orthopaedic surgical oncology
Introduction
Total joint arthroplasties (TJAs) are highly successful orthopaedic surgical interventions and are most frequently applied in patients with degenerative joint disorders. The number of TJAs being implanted annually is still rising due to an ageing population. It is estimated that approximately four million TJAs will be performed in 2030 in the USA alone (1). Even though these procedures are usually successful with substantial improvement in the patient’s quality of life, 1–2% of patients with primary implants and 7% of patients with revised implants develop periprosthetic joint infection (PJI) (2). Although the prevalence of PJI is relatively low, its clinical impact is evident. Due to formation of a biofilm at the surface of the implant it is hard to treat PJI. This results in the need for extensive surgical re-interventions, which is accompanied by high morbidity (3).
As with TJAs indicated for degenerative joint disorders, PJI is also reported in patients with oncologic TJAs [periprosthetic infection of a megaprosthesis (PMI)]. The prevalence of PMI after oncologic TJA is much higher than after regular TJA and ranges from 7% to 28% (4-6). This higher prevalence is reasonably caused by local and systemic immunodeficiency, long duration of surgery, large wound areas and the use of large implants due to extensive tumor resection (7,8). Oncology patients are often immunodeficient because of the application of (neoadjuvant) chemotherapy or radiotherapy. Immunodeficiency leads to a decreased host defense, increased bleeding and compromised soft tissue healing. Moreover, due to prolonged duration of the procedure and a larger implant, the orthopeadic oncology patient has an even higher risk of compromised soft tissue healing. In addition, larger wound areas and resection could result in significant dead space and subsequently massive hematoma formation which further increases the risk of infection (9).
In order to assess the risk of infection after oncologic TJA this article focuses on specific issues regarding PJI in orthopaedic oncological implant surgery, aiming to give directions for the prevention, diagnosis and treatment of PMI.
Prevention
Antibiotic prophylaxis
Perioperative prophylactic antibiotic treatment has proven to be effective in reducing the risk of PJI after regular TJA from 5% to 1% when one preoperative dose and two postoperative doses of prophylactic antibiotics are administered within the first 24 hours after TJA (10). In oncologic TJA it is clinical practice in many hospitals to administer prophylactic antibiotics longer than 24 hours after TJA (up to 5 days). This is in line with the conclusions of a systematic review conducted by Racano et al. which showed that extended postoperative antibiotic prophylaxis can reduce the risk of PMI from 13% to 8% (8). Limitations of this review are the variety and inconsistency of antibiotics administered, lack of a clear definition of PMI, and low level of evidence of the included studies (8). These results emphasize the heterogeneity in the orthopaedic oncology patients compared to conventional orthopaedic patients, which indicates that various antibiotic prophylaxis strategies may be required. Thereby, it demonstrates the importance of composing evidence-based guidelines for antibiotic prophylaxis in oncologic orthopaedic surgery. Currently, an international multicentre study is performed to compare the effect of prophylactic antibiotic duration of 24 hours versus 5 days after oncologic TJA (PARITY study) (11). The results of this study may provide sufficient evidence on the appropriate duration of antibiotic prophylaxis.
A different method of administering antibiotic prophylaxis is soaking the endoprosthesis and/or allograft in antibiotic or antiseptic solutions before implantation. This soaking could result in a reduced infection rate by releasing local antimicrobials and thereby diminishing the bacterial load on the implant prior to implantation. However, only few studies have been conducted to evaluate the effectiveness of this type of prophylaxis. Therefore, implementation of soaking as standard antibiotic prophylaxis remains debatable (12).
Endoprosthetic coatings
The use of endoprosthetic coatings is an effective preventive measure for PMI. Endoprosthetic coatings provide a technique for the local delivery of antibiotics or antiseptics and show promising results in in vitro studies (13). These coatings can prevent bacterial colonization and promote ingrowth of the TJA, which is especially important in the application of cementless implants. Recent studies investigated the efficacy of silver coatings (14). Silver coatings possess bactericidal properties operating by disrupting the cellular membrane of microorganisms and preventing DNA formation. Several studies reported a decrease of 50% in the prevalence of PMI when using a silver coating compared to uncoated TJAs (15,16). Hardes et al. showed that silver levels remained present in the serum up to 24 months after implantation of the silver coated TJA, but despite this finding no adverse local tissue reactions or systemic toxicity were reported (17). Bosetti et al. determined the silver concentration in drains and seroma in patients with a silver coated megaprosthesis. Local argyria occurred in 23% of patients, independent of the silver concentration. No systemic toxic reactions to silver have been reported (18). Although results regarding silver coated prosthesis are promising, additional prospective studies are needed to confirm the effectivity of silver coated TJAs in oncology patients.
Definition and classification
There are several guidelines that can be used for the definition and classification of PMI. The most commonly used definitions for general PJI are based on the guidelines developed by the Infectious Diseases Society of America (IDSA) and Musculoskeletal Infection Society (MSIS) (19,20). These general PJI definitions can also be applied to PMI. It is advised to use one of these two definitions, so that the results in literature are comparable and can be used for the estimation of the prognosis and success of treatment strategies.
Diagnosis
Regarding the diagnosis of PMI, similar principles apply as for general PJI. The suspicion of PMI can be raised based on symptoms, radiological signs, serological markers and synovial markers. Regarding the symptoms of PMI, pain at the site of implant is a consistent symptom of infection in over 90% of cases of PMI (21). As for radiological signs, the following signs indicate PMI: a periosteal reaction, scattered foci of osteolysis, or generalized bone resorption in the absence of implant wear (22).
Various serological markers are available for diagnostic screening. The most commonly used markers are C-reactive protein (CRP), erythrocyte sedimentation rate (ESR) and white blood cell count (WBC). The combined use of CRP and ESR can be helpful in the diagnosis of PMI, since their combined sensitivity and specificity are 99% and 89%, respectively (23). After uncomplicated TJA the serum CRP level increases rapidly within 2 to 3 days, with normalization to the preoperative level in 2 to 8 weeks (24,25). The ESR increases more gradually than the CRP, with peak level between day 5 and 14 and normalization in up to 9 months after TJA (24,26). PMI should be suspected if CRP levels increase later than 3 days after TJA or remain elevated (25,27). Nevertheless, increased values of these serological markers may also be associated with oncologic conditions, which decreases their usefulness in oncology patients (28). Moreover, WBC may be reduced in oncology patients as a result of myelosuppressive chemotherapy.
If PMI is suspected based on clinical symptoms, radiological signs or serological markers, joint aspiration should be performed. There are several synovial markers that can be used, of which CRP, WBC and percentage of neutrophils are most frequently used. Synovial CRP shows sensitivity 70–97% and specificity 79–100% (29,30), synovial WBC shows sensitivity 36–100% and specificity 80–99% (31,32), and percentage of neutrophils shows sensitivity 84–100% and specificity 80–99% (31,32). The sensitivity can be improved by serial aspirations. Currently, other synovial biomarkers are being tested with good results, such as leukocyte esterase (33), alpha-defensin (34), and calprotectin (35). When PMI is suspected but cannot be confirmed by serological or synovial markers, bone scintigraphy, white blood cell scan or PET-scan can be performed.
The golden standard for diagnosing PMI is obtaining positive intra-articular cultures. Before obtaining these cultures, antibiotics should be withheld for a minimum of 2 weeks, to minimize the risk of false-negative results. Nevertheless, a single dose of prophylactic antibiotics does not alter intra-articular culture results and should therefore not be withheld (36). The incubation period of cultures should be at least 7 days, although a minimum of 2 weeks is advised (37). The most common microorganisms causing PMI are Staphylococcus aureus and coagulase negative staphylococci. Together they account for over 50% of PMIs. A large proportion of PMIs are polymicrobial infections (21–45%) (5,38).
Treatment
DAIR versus revision surgery
Debridement, antibiotics and implant retention (DAIR) is one of the treatment strategies frequently applied for PMI in orthopaedic oncology patients, especially in early PMI (within 3 months after index surgery) (39,40). Nevertheless, infection control after DAIR varies widely with success rates varying from 39% to 70% (15,41,42). Several factors are associated with a higher rate of infection control after DAIR, including: early infection, short duration of symptoms, well-fixed implants and pre-operatively identified microorganisms (43). Unfortunately, studies studies reporting on the effectiveness of DAIR in oncology patients are scarce and have large variation in follow-up periods. In our opinion, DAIR should be used as the first treatment option in early PMI, especially considering the enormous consequences of removal of the large arthroplasty in oncology patients.
If DAIR fails to eradicate the infection, two-stage revision surgery is usually necessary. In literature, high rates of infection control are reported for two-stage revision surgery, with success rates varying from 63% to 100% (4,38,44). Other treatment strategies include one-stage revision, suppressive antibiotics and amputation, but all are considered less appropriate as initial treatment for PMI, most importantly since the rates of infection control after one-stage revision are worse than after two-stage revision (5,38,45). In addition, rates of infection control are higher when DAIR and two-stage revisions are performed in patients with a silver coated TJA (15).
Use of a spacer in two-stage revision surgery
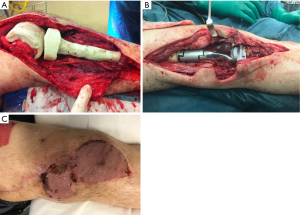
The use of antibiotic-loaded cement spacers in two-stage revision surgery offers the advantages of delivering antibiotics locally at the site of infection while maintaining soft tissue tension to facilitate reimplantation of the TJA. In literature, no details are published on the design or composition of antibiotic-loaded cement spacers in case of two-stage revision surgery in PMI. Therefore, at this time point the current concepts of the use of spacers in two-stage revision in regular PJI should be applied to the oncology patients as well. According to these concepts, antibiotics added to the cement spacer should be heat-stable and should provide coverage for the identified or suspected microorganisms causing the infection (46). Figure 1A illustrates the application of a customized spacer during the first stage of two-stage revision surgery. If the soft tissue is of acceptable quality at the time of surgery, a two-component spacer is a good option to prevent stiffness of the joint. Figure 1B illustrates reimplantation of the implant during the second stage of revision surgery. Figure 1C shows that a gastrocnemius muscle flap with free skin graft is used to cover the soft tissue defect. This indicates that it is valuable to collaborate with a plastic surgeon to provide good soft tissue coverage in two-stage revision surgery, especially in reconstructions in the proximal tibia and distal femur.
Use of antibiotics in the treatment of PMI
Literature regarding the use of antibiotics in the surgical treatment of PMI is scarce. In the available literature on oncology patients, antibiotic regimens differed per patient according to the individual culture results and local protocol, about which no details were provided. Therefore, at this time point the current concepts for the use of antibiotics in regular PJI should be applied to oncology patients as well. In general, antibiotics are administered intravenously for at least 2 weeks, followed by oral antibiotics for at least 10 weeks. The type of antibiotics is adjusted according to the culture results and resistance patterns, in consultation with an infectious diseases specialist.
Influence of radiotherapy and chemotherapy on infection control
Radiotherapy increases the risk of PMI (45,47). Also, it increases the rate of failure after two-stage revision for PMI (4,44). Postoperative radiotherapy is of bigger influence on the infection rate than preoperative radiotherapy. This especially accounts for DAIR procedures, in which postoperative radiotherapy negatively influences the quality of the soft tissues and therefore hampers the local defence mechanisms and local availability of antibiotics (47).
Several studies found an increased rate of PMI in patients receiving chemotherapy with incidences ranging from 18% to 32% (15,48,49), although other studies could not confirm these results (5,43,50). In patients receiving two-stage revision surgery, it is advised to delay reimplantation of the TJA until completion of chemotherapy, because of the immunocompromised status of oncology patients receiving chemotherapy and the associated high-risk regarding re-infection (43).
Conclusions
While the diagnosis and treatment of regular PJI are already challenging, PMI poses additional challenges because of the immunocompromised status of oncology patients and extensive tumor resections. In general, regular PJI guidelines can be used for the prevention, diagnosis and treatment of PMI, although differences between regular patients and oncology patients should be kept in mind. Most importantly, wound healing after TJA should be monitored intensively, especially in patients receiving radiotherapy or chemotherapy, since these patients are immunocompromised and have a diminished quality of soft tissue surrounding the joint. Perioperative antibiotic prophylaxis could be extended in these patients to reduce the risk of PMI. Current ongoing studies may provide clear recommendations for this in the future. Furthermore, silver coated TJAs could provide beneficial effects in the prevention of PMI. In case PMI has developed, DAIR is the first choice of surgical treatment, especially in early infections. Two-stage revision surgery is a viable option in case DAIR is unsuccessful. Reimplantation of the TJA should be postponed until chemotherapy is completed.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Annals of Joint for the series “Reconstruction in Orthopaedic Oncology - Frontier and Future Trends”. The article has undergone external peer review.
Conflict of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2019.05.01). The series “Reconstruction in Orthopaedic Oncology - Frontier and Future Trends” was commissioned by the editorial office without any funding or sponsorship. PCJ served as the unpaid Guest Editor of the series. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Darouiche RO. Treatment of infections associated with surgical implants. N Engl J Med 2004;350:1422-9. [Crossref] [PubMed]
- Kurtz SM, Ong KL, Lau E, et al. Prosthetic joint infection risk after TKA in the medicare population. Clin Orthop Relat Res 2010;468:52-6. [Crossref] [PubMed]
- Abad CL, Haleem A. Prosthetic joint infections: an update. Curr Infect Dis Rep 2018;20:15. [Crossref] [PubMed]
- Grimer RJ, Belthur M, Chandrasekar C, et al. Two-stage revision for infected endoprostheses used in tumor surgery. Clin Orthop Relat Res 2002;193-203. [Crossref] [PubMed]
- Hardes J, Gebert C, Schwappach A, et al. Characteristics and outcome of infections associated with tumor endoprostheses. Arch Orthop Trauma Surg 2006;126:289-96. [Crossref] [PubMed]
- Jeys LM, Grimer RJ, Carter SR, et al. Post operative infection and increased survival in osteosarcoma patients: are they associated? Ann Surg Oncol 2007;14:2887-95. [Crossref] [PubMed]
- Nagano S, Yokouchi M, Setoguchi T, et al. Analysis of surgical site infection after musculoskeletal tumor surgery: risk assessment using a new scoring system. Sarcoma 2014;2014:645496 [Crossref] [PubMed]
- Racano A, Pazionis T, Farrokhyar F, et al. High infection rate outcomes in long-bone tumor surgery with endoprosthetic reconstruction in adults: a systematic review. Clin Orthop Relat Res 2013;471:2017-27. [Crossref] [PubMed]
- Kapoor SK, Thiyam R. Management of infection following reconstruction in bone tumors. J Clin Orthop Trauma 2015;6:244-51. [Crossref] [PubMed]
- Yates AJ. Postoperative prophylactic antibiotics in total joint arthroplasty. Arthroplast Today 2018;4:130-1. [Crossref] [PubMed]
- Ghert M, Deheshi B, Holt G, et al. Prophylactic antibiotic regimens in tumour surgery (PARITY): protocol for a multicentre randomised controlled study. BMJ Open 2012;2:e002197 [Crossref] [PubMed]
- Berríos-Torres SI, Umscheid CA, Bratzler DW, et al. Centers for disease control and prevention guideline for the prevention of surgical site infection, 2017. JAMA Surg 2017;152:784-91. [Crossref] [PubMed]
- Neut D, Dijkstra RJB, Thompson JI, et al. Antibacterial efficacy of a new gentamicin-coating for cementless prostheses compared to gentamicin-loaded bone cement. J Orthop Res 2011;29:1654-61. [Crossref] [PubMed]
- Romanò CL, Scarponi S, Gallazzi E, et al. Antibacterial coating of implants in orthopaedics and trauma: a classification proposal in an evolving panorama. J Orthop Surg Res 2015;10:157. [Crossref] [PubMed]
- Wafa H, Grimer R, Reddy K, et al. Retrospective evaluation of the incidence of early periprosthetic infection with silver-treated endoprostheses in high-risk patients: case-control study. Bone Joint J 2015;97-B:252-7. [Crossref] [PubMed]
- Donati F, Di Giacomo G, D’Adamio S, et al. Silver-coated hip megaprosthesis in oncological limb savage surgery. Biomed Res Int 2016;2016:9079041 [Crossref] [PubMed]
- Hardes J, Ahrens H, Gebert C, et al. Lack of toxicological side-effects in silver-coated megaprostheses in humans. Biomaterials 2007;28:2869-75. [Crossref] [PubMed]
- Bosetti M, Massè A, Tobin E, et al. Silver coated materials for external fixation devices: in vitro biocompatibility and genotoxicity. Biomaterials 2002;23:887-92. [Crossref] [PubMed]
- Parvizi J, Zmistowski B, Berbari E, et al. New definition for periprosthetic joint infection: from the workgroup of the musculoskeletal infection society. Clin Orthop Relat Res 2011;469:2992-4. [Crossref] [PubMed]
- Osmon DR, Berbari EF, Berendt AR, et al. Diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 2013;56:e1-e25. [Crossref] [PubMed]
- Tsaras G, Osmon D, Mabry T, et al. Incidence, secular trends and outcomes of prosthetic joint infection (PJI): a population based study, Olmsted county, Minnesota, 1969-2007. Infect Control Hosp Epidemiol 2012;33:1207-12. [Crossref] [PubMed]
- Ostlere S. How to image metal-on-metal prostheses and their complications. AJR Am J Roentgenol 2011;197:558-67. [Crossref] [PubMed]
- Costa CR, Johnson AJ, Naziri Q, et al. Efficacy of erythrocyte sedimentation rate and C-reactive protein level in determining periprosthetic hip infections. Am J Orthop 2012;41:160-5. [PubMed]
- Bilgen O, Atici T, Durak K, et al. C-reactive protein values and erythrocyte sedimentation rates after total hip and total knee arthroplasty. J Int Med Res 2001;29:7-12. [Crossref] [PubMed]
- White J, Kelly M, Dunsmuir R. C-reactive protein level after total hip and knee replacement. J Bone Joint Surg Br 1998;80:909-11. [Crossref] [PubMed]
- Honsawek S, Deepaisarnsakul B, Tanavalee A, et al. Relationship of serum IL-6, C-reactive protein, erythrocyte sedimentation rate, and knee skin temperature after total knee arthroplasty: a prospective study. Int Orthop 2011;35:31-5. [Crossref] [PubMed]
- Niskanen RO, Korkala O, Pammo H. Serum C-reactive protein levels after total hip and knee arthroplasty. J Bone Joint Surg Br 1996;78:431-3. [Crossref] [PubMed]
- Van Hemelrijck M, Holmberg L, Garmo H, et al. Association between levels of C-reactive protein and leukocytes and cancer: Three repeated measurements in the Swedish AMORIS study. Cancer Epidemiol Biomarkers Prev 2011;20:428-37. [Crossref] [PubMed]
- Parvizi J, Jacovides C, Adeli B, et al. Synovial C-reactive protein: a prospective evaluation of a molecular marker for periprosthetic joint infection. Clin Orthop Relat Res 2012;470:54-60. [Crossref] [PubMed]
- Tetreault MW, Wetters NG, Moric M, et al. Is synovial C-reactive protein a useful marker for periprosthetic joint infection? Clin Orthop Relat Res 2014;472:3997-4003. [Crossref] [PubMed]
- Dinneen A, Guyot A, Clements J, et al. Synovial fluid white cell and differential count in the diagnosis or exclusion of prosthetic joint infection. Bone Joint J 2013;95-B:554-7. [Crossref] [PubMed]
- Ghanem E, Parvizi J, Burnett R, et al. Cell count and differential of aspirated fluid in the diagnosis of infection at the site of total knee arthroplasty. J Bone Joint Surg Am 2008;90:1637-43. [Crossref] [PubMed]
- Wetters NG, Berend KR, Lombardi AV, et al. Leukocyte esterase reagent strips for the rapid diagnosis of periprosthetic joint infection. J Arthroplasty 2012;27:8-11. [Crossref] [PubMed]
- Deirmengian C, Kardos K, Kilmartin P, et al. The alpha-defensin test for periprosthetic joint infection outperforms the leukocyte esterase test strip. Clin Orthop Relat Res 2015;473:198-203. [Crossref] [PubMed]
- Wouthuyzen-Bakker M, Ploegmakers J, Kampinga G, et al. Synovial calprotectin: a potential biomarker to exclude a prosthetic joint infection. Bone Joint J 2017;99-B:660-5. [Crossref] [PubMed]
- Wouthuyzen-Bakker M, Benito N, Soriano A. The effect of preoperative antimicrobial prophylaxis on intraoperative culture results in patients with suspected or confirmed prosthetic joint infection: a systematic review. J Clin Microbiol 2017;55:2765-74. [Crossref] [PubMed]
- Fink B, Makowiak C, Fuerst M, et al. The value of synovial biopsy, joint aspiration and C-reactive protein in the diagnosis of late peri-prosthetic infection of total knee replacements. J Bone Joint Surg Br 2008;90:874-8. [Crossref] [PubMed]
- Jeys LM, Grimer RJ, Carter SR, et al. Periprosthetic infection in patients treated for an orthopaedic oncological condition. J Bone Joint Surg Am 2005;87:842-9. [Crossref] [PubMed]
- Yokoe DS, Avery TR, Platt R, et al. Reporting surgical site infections following total hip and knee arthroplasty: impact of limiting surveillance to the operative hospital. Clin Infect Dis 2013;57:1282-8. [Crossref] [PubMed]
- Barrett L, Atkins B. The clinical presentation of prosthetic joint infection. J Antimicrob Chemother 2014;69:i25-7. [Crossref] [PubMed]
- Dhanoa A, Ajit Singh V, Elbahri H. Deep infections after endoprosthetic replacement operations in orthopedic oncology patients. Surg Infect (Larchmt) 2015;16:323-32. [Crossref] [PubMed]
- Peel TN, Cheng AC, Buising KL, et al. Microbiological aetiology, epidemiology, and clinical profile of prosthetic joint infections: are current antibiotic prophylaxis guidelines effective? Antimicrob Agents Chemother 2012;56:2386-91. [Crossref] [PubMed]
- Mavrogenis AF, Papagelopoulos PJ, Coll-Mesa L, et al. Infected tumor prostheses. Orthopedics 2011;34:991-8. [Crossref] [PubMed]
- Flint MN, Griffin AM, Bell RS, et al. Two-stage revision of infected uncemented lower extremity tumor endoprostheses. J Arthroplasty 2007;22:859-65. [Crossref] [PubMed]
- Funovics PT, Hipfl C, Hofstaetter JG, et al. Management of septic complications following modular endoprosthetic reconstruction of the proximal femur. Int Orthop 2011;35:1437-44. [Crossref] [PubMed]
- Bloomfield MR, Klika AK, Barsoum WK. Antibiotic-coated spacers for total hip arthroplasty infection. Orthopedics 2010;33:649. [PubMed]
- Jeys LM, Luscombe JS, Grimer RJ, et al. The risks and benefits of radiotherapy with massive endoprosthetic replacement. J Bone Joint Surg Br 2007;89:1352-5. [Crossref] [PubMed]
- Lee SH, Oh JH, Lee KS, et al. Infection after prosthetic reconstruction in limb salvage surgery. Int Orthop 2002;26:179-84. [Crossref] [PubMed]
- McDonald DJ, Capanna R, Gherlinzoni F, et al. Influence of chemotherapy on perioperative complications in limb salvage surgery for bone tumors. Cancer 1990;65:1509-16. [Crossref] [PubMed]
- Morii T, Yabe H, Morioka H, et al. Postoperative deep infection in tumor endoprosthesis reconstruction around the knee. J Orthop Sci 2010;15:331-9. [Crossref] [PubMed]
Cite this article as: Zuidhof RJWJ, Löwik CAM, Ploegmakers JJW, Dijkstra SPD, Wouthuyzen-Bakker M, Jutte PC. Periprosthetic joint infection in orthopaedic surgical oncology. Ann Joint 2019;4:26.


