Anatomical anterior cruciate ligament reconstruction with a flat graft using a new tunnel creation technique
Introduction
Motivated by the development of double bundle anterior cruciate ligament (ACL) reconstruction the interest in ACL anatomy led to the discovery of its flat femoral direct insertion and flat intraligamentous structure (1-4). The picture was completed by the anatomical description of Smigielski et al. (5). According to his findings (5-7) and those of others (3,8-10) the whole intraligamentous ACL appears to be flat, “ribbon-like” after removal of the synovial membrane. Smigielski et al. also rediscovered the tibial relationship between the C-shaped tibial attachment of the ACL and the lateral meniscus described by Testut und Jacob in 1921 (7).
By placing the graft in round Single Bundle (SB) bone tunnels the structure and composition of the direct insertion site is not reproduced. The principle of the new introduced “Medacta Anatomic Ribbon Surgery” (M-ARS) (Medacta International, Switzerland) technique is to create rectangular bone slits that resemble more closely the original flat ACL insertions on the femur and tibia to allow for a flat ACL reconstruction.
Biomechanical in vivo studies found that a flat graft alignment (as approximated DB ACL reconstruction) is advantageous in restoring rotational kinematics and stability compared to SB, when referenced to the healthy contralateral knee (11,12). Comparable kinematics to a native ACL could also be shown in ACL reconstruction using rectangular bone slits (13-15).
The new M-ARS technique could also potentially provide interesting biological benefits. As the contact area of a flat ACL graft is about 3 times greater in relation to its volume compared to a round one, the formation of Sharpey-like fibers as well as the vascularization can take place on a larger area. This may reduce central necrosis as described in the early graft healing phase (16) due to a reduced distance for diffusion.
The biomechanical and biological aims of the new ACL reconstruction technique are:
- Reconstruction of a straight rectangular bone slit for the femoral insertion;
- Reconstruction of a curved c-shaped tibial bone slit with preservation of the anterior horn of the lateral meniscus and the possibility of a more anterior (anatomical) position without notch impingement;
- Use of a flat tendon graft which better fits in the notch without reducing total graft volume and resembling the native ACL with different fiber recruitment under tension throughout the range of motion;
- Increased interface for tendon to bone healing with potentially improved or accelerated revascularization and tendon to bone healing.
Technique
Graft harvesting
Hamstring tendon or Quadriceps tendon (QT) are ideal for this flat M-ARS ACL reconstruction and may be used according to surgeons’ preference.
Steps for graft preparation Semitendinosus tendon
- A length of 26 cm for a 4-fold Semitendinosus tendon (Semi-T) graft is needed.
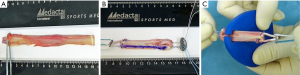
- The tendon is cleaned from muscle tissue and is placed with the muscular side upwards on the preparation board. Starting from the proximal (muscular) end, the tendon is split with a knife towards the round distal end (pes anserinus). Then the tendon is flattened with a forceps to create an equal flat dimension from end to end (Figure 1A).
- The flat tendon is folded in the middle to create a 2-fold graft. For proximal fixation it is folded a second time over the loop of a (adjustable/flexible) suture button to create a 4-fold graft (Figure 1B).
- If using a fixed femoral loop button, it must be chosen at the appropriate length to fit the femoral bone slit length. An easier way is to use an adjustable/flexible loop button. At least 15 mm of the flat graft should be inserted into the femoral (and tibial) slit. The length of the femoral tunnel should be marked on the graft.
- For distal fixation the tendinous end of the graft is prepared using an interlocking suturing technique (Krackow-stitch) with two No. 2 non-absorbable sutures, but without the sutures in the mid part. Then the distal sutures are thread through the tibial fixation button such that the concavity of the button looks to the lateral graft side (Figure 1B).
- The size of the graft should be measured to define the final dimensions of the bone slits with the flat measuring device (S-small, M-medium, L-large) (Figure 1C).
Quadriceps tendon
QT may be harvested in a conventional open technique or with a minimal invasive technique using special harvesting instrumentation (17). The graft should have a thickness of about 4.5 mm, a width of 10–12 mm and a length of a minimum of 6.5–7 cm. Over the patella the periosteal strip of about 2 cm of the QT is carefully elevated and then cut (17).
Graft preparation Quad tendon
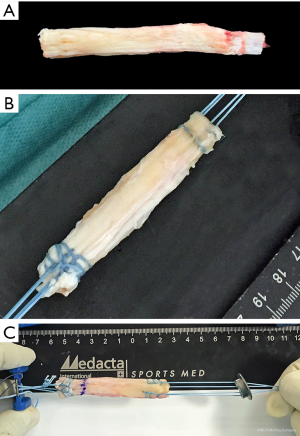
- The distal end of the graft (periosteum) (Figure 2A), which will become the femoral graft end, is folded over a suture that is removed after preparation and kept in place with a Lahey-goiter grasping forceps.
- Krackow-stitch sutures (Figure 2B) are placed on each side of the graft using non-absorbable sutures size 2. The stitches should be done in the outer 3 mm of the graft and the sutures should go back in the mid part of the graft using a simple spiral seem to get pulling strands in every portion of the proximal graft edge.
- Putting tension on all sutures shows a slight C-shape form of the proximal graft end and either the concavity or convexity should be marked as lateral or medial, respectively, for insertion.
- The tendinous end of the graft, which will become the tibial end of the implanted graft, is prepared similar to Semi-T (Figure 2C).
Femoral slit
- The ACL remnants are removed keeping the femoral attachment and the C-shaped tibial one (5).
- In 110° of flexion, a central guide wire (with a laser mark is drilled into the mid portion of the native femoral ACL insertion using a medial portal technique. The entry point is before marked with a microfracture ale after measuring the length of the attachment with a ruler. The central wire is introduced until the laser mark is flush with the femoral condyle and the position is double-checked from the medial portal. Now the tunnel length is measured with an outside-in measurement device from extra-articular.
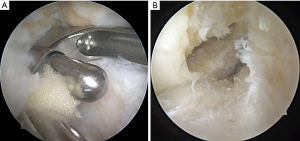
- The femoral aimer is inserted into the joint and over the central guide wire using the central hole. Its orientation should be in line with the ACL attachment, which is approximately horizontal in 110° of knee flexion (4) (Figure 3A).
- The anterior and posterior guide wires are drilled through the two peripheral holes of the femoral aimer, approximately 25–30 mm deep.
- The femoral aimer is removed and the peripheral two wires are over-drilled with a 4.5 mm drill to a depth of 25–30 mm depending on the length of the central femoral tunnel without over-drilling the lateral cortex. The central guide wire is over-drilled through the lateral cortex with the 4.5 mm drill and is left in place.
- A femoral dilator matching the graft size (S, M, L) is inserted over the central guide wire and tapped in to a depth of 25–30 mm. The resulting slit should be 10 mm deeper than the length of the graft in the femoral slit to allow the femoral fixation button to flip (Figure 3B).
Tibial slit
- The tibial aimer is chosen according to the side (r/l) and size of the graft (S, M, L). The threaded central k-wire is inserted into the tip hole of the bullet and is inserted into the tibial aimer.
- The aimer is inserted through the medial portal with the knee in 90° of flexion and placed around the anterior horn of the lateral meniscus and with reference to the ACL remnant. The threaded central k-wire is drilled into the joint (Figure 4A).
- First the shorter 4.5 mm drill bit is drilled through the anterior hole of the tibial aimer until just breaking the joint cortex and left in place (Figure 4A). Then the longer 4.5 mm drill bit is drilled through the posterior hole in a similar way. Both 4.5 mm drills and the aimer is removed leaving the threaded central k-wire.
- It is now overdrilled by the cannulated 4.5 mm drill. Then everything is removed.
- The C-shape slit is finally prepared with the special dilatator using the two blunt guide pins in the anterior and posterior tibial 4.5 mm tunnel (Figure 4B). The size of the dilatator should be chosen according to S/M/L and it should be oriented with its concavity to the lateral meniscus. The distal part of the c-shaped slit should be dilated with the large dilator for perfect fit of the tibial fixation button.
Graft insertion
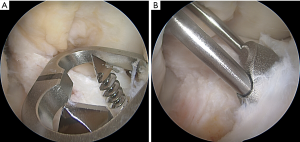
- The graft is pulled into position from distal to proximal through the tibial slit in the usual way. Correct orientation within the tibial tunnel is with the concavity looking laterally (Figure 5A).
- Once the graft emerges from the proximal tibial tunnel into the joint, the knee should be extended to get proper torque of the graft. Alternatively, the proximal end of the graft should be twisted for proper anatomical tibial-to-femoral fiber alignment (AM to AM, PM to PM) before pulling it into the femoral bone slit and before flipping of the femoral button is performed (Figure 5A).
- Finally, the knee is cycled to condition the graft and tibial fixation is performed with the tibial fixation button close to extension (Figure 5B).
Postoperative care
The patient is mobilized with partial weight-bearing (15–20 kg) for 1–2 week, followed by full weight-bearing. Free range of motion is started directly postoperatively. Physical therapy is recommended 2–3 times per week.
Discussion
A new concept of single bundle ACL reconstruction is presented using either a Semi-T- or quadriceps tendon graft. The flat intratendinous alignment of the graft appears to be close to the native ACL anatomy (5-7). As any ACL surgery the new M-ARS ACL reconstruction technique has some pearls and pitfalls as well as some advantages and disadvantages compared to the conventional technique.
Our conventional technique for ACL reconstruction is to use drills to create bone tunnels for graft insertion. However, neither the femoral nor the tibial anatomical insertion sites are round—in contrast—flat and C-shaped tunnels respectively may offer obvious and potential advantages.
On the femoral side the creation of a flat slit mimics the direct attachment of the ACL and the introduction of a flat graft resembles the native intraligamentous “ribbon-like” ACL, changing its fiber orientation according to different flexion angles without overstuffing the notch.
The native tibial insertion site has been shown to be C- (67%) or J-shaped (24%) (5), which can be achieved most closely with the C-shaped slit in the tibia. The C-shaped slit around the root of the anterior horn of the lateral meniscus potentially avoids laceration of the lateral meniscus compared to conventional round tunnel placement (18-20).
In addition, the increased graft -bone contact area of a flat ACL graft in a bone slit compared to a round graft with similar cross-sectional area may provide a biological advantage leading to accelerated ingrowth and probably less central graft necrosis. The flat graft appearance may also present biomechanical advantages similar to ACL double bundle reconstruction or rectangular bone tunnels (13-15,21-24).
Conclusions
A new concept of single bundle ACL reconstruction appears to be close to the native ACL anatomy. As any ACL surgery, the new M-ARS ACL reconstruction technique has advantages and disadvantages. However early clinical experiences of the new technique are very promising with little risk for complications. Controlled clinical trials have to be conducted to evaluate whether the new technique leads to better mid-term to long-term results and could be beneficial for patients.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Takeshi Muneta) for the series “Anatomic Reconstruction of Anterior Cruciate Ligament - Concept, Indication, and Its Efficacy” published in Annals of Joint. The article has undergone external peer review.
Conflict of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2019.05.02). The series “Anatomic Reconstruction of Anterior Cruciate Ligament - Concept, Indication, and Its Efficacy” was commissioned by the editorial office without any funding or sponsorship. RS reports personal fees from Medacta International, outside the submitted work and has a patent Medacta International with royalties paid. RŚ reports personal fees from Medacta International, outside the submitted work and has a patent Medacta International with royalties paid. MH reports personal fees from Medacta International, personal fees from Conmed Deutschland GmbH, personal fees from DJO - Ormed GmbH, personal fees from Arthrex, personal fees from OPED, outside the submitted work. In addition, MH has a patent Medacta International with royalties paid. CF reports personal fees from Medacta International, personal fees from Karl Storz, outside the submitted work and has a patent Medacta International with royalties paid. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Iwahashi T, Shino K, Nakata K, et al. Direct anterior cruciate ligament insertion to the femur assessed by histology and 3-dimensional volume-rendered computed tomography. Arthroscopy 2010;26:S13-20. [Crossref] [PubMed]
- Mochizuki T, Fujishiro H, Nimura A, et al. Anatomic and histologic analysis of the mid-substance and fan-like extension fibres of the anterior cruciate ligament during knee motion, with special reference to the femoral attachment. Knee Surg Sports Traumatol Arthrosc 2014;22:336-44. [Crossref] [PubMed]
- Mochizuki T, Muneta T, Nagase T, et al. Cadaveric knee observation study for describing anatomic femoral tunnel placement for two-bundle anterior cruciate ligament reconstruction. Arthroscopy 2006;22:356-61. [Crossref] [PubMed]
- Siebold R, Ellert T, Metz S, et al. Femoral insertions of the anteromedial and posterolateral bundles of the anterior cruciate ligament: morphometry and arthroscopic orientation models for double-bundle bone tunnel placement--a cadaver study. Arthroscopy 2008;24:585-92. [Crossref] [PubMed]
- Smigielski R, Zdanowicz U, Drwiega M, et al. The anatomy of the anterior cruciate ligament and its relevance to the technique of reconstruction. Bone Joint J 2016;98-b:1020-6.
- Siebold R, Schuhmacher P, Fernandez F, et al. Flat midsubstance of the anterior cruciate ligament with tibial "C"-shaped insertion site. Knee Surg Sports Traumatol Arthrosc 2015;23:3136-42. [Crossref] [PubMed]
- Śmigielsk R, Zdanowicz U, Drwiega M, et al. Ribbon like appearance of the midsubstance fibres of the anterior cruciate ligament close to its femoral insertion site: a cadaveric study including 111 knees. Knee Surg Sports Traumatol Arthrosc 2015;23:3143-50. [Crossref] [PubMed]
- Kawaguchi Y, Kondo E, Takeda R, et al. The role of fibers in the femoral attachment of the anterior cruciate ligament in resisting tibial displacement. Arthroscopy 2015;31:435-44. [Crossref] [PubMed]
- Nawabi DH, Tucker S, Schafer KA, et al. ACL Fibers Near the Lateral Intercondylar Ridge Are the Most Load Bearing During Stability Examinations and Isometric Through Passive Flexion. Am J Sports Med 2016;44:2563-71. [Crossref] [PubMed]
- Oka S, Schuhmacher P, Brehmer A, et al. Histological analysis of the tibial anterior cruciate ligament insertion. Knee Surg Sports Traumatol Arthrosc 2016;24:747-53. [Crossref] [PubMed]
- Logan MC, Williams A, Lavelle J, et al. Tibiofemoral kinematics following successful anterior cruciate ligament reconstruction using dynamic multiple resonance imaging. Am J Sports Med 2004;32:984-92. [Crossref] [PubMed]
- Ristanis S, Giakas G, Papageorgiou CD, et al. The effects of anterior cruciate ligament reconstruction on tibial rotation during pivoting after descending stairs. Knee Surg Sports Traumatol Arthrosc 2003;11:360-5. [Crossref] [PubMed]
- Herbort M, Tecklenburg K, Zantop T, et al. Single-bundle anterior cruciate ligament reconstruction: a biomechanical cadaveric study of a rectangular quadriceps and bone--patellar tendon--bone graft configuration versus a round hamstring graft. Arthroscopy 2013;29:1981-90. [Crossref] [PubMed]
- Jacobi M, Magnussen RA, Villa V, et al. The concept of double bundle ACL simulation with a single bundle patellar tendon graft. A cadaveric feasibility study. Sports Med Arthrosc Rehabil Ther Technol 2012;4:19. [Crossref] [PubMed]
- Shino K, Nakata K, Nakamura N, et al. Rectangular tunnel double-bundle anterior cruciate ligament reconstruction with bone-patellar tendon-bone graft to mimic natural fiber arrangement. Arthroscopy 2008;24:1178-83. [Crossref] [PubMed]
- Janssen RP, Scheffler SU. Intra-articular remodelling of hamstring tendon grafts after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 2014;22:2102-8. [Crossref] [PubMed]
- Fink C, Herbort M, Abermann E, et al. Minimally invasive harvest of a quadriceps tendon graft with or without a bone block. Arthrosc Tech 2014;3:e509-13. [Crossref] [PubMed]
- Furumatsu T, Kodama Y, Maehara A, et al. The anterior cruciate ligament-lateral meniscus complex: A histological study. Connect Tissue Res 2016;57:91-8. [Crossref] [PubMed]
- Kodama Y, Furumatsu T, Miyazawa S, et al. Location of the tibial tunnel aperture affects extrusion of the lateral meniscus following reconstruction of the anterior cruciate ligament. J Orthop Res 2017;35:1625-33. [Crossref] [PubMed]
- Kusano M, Yonetani Y, Mae T, et al. Tibial insertions of the anterior cruciate ligament and the anterior horn of the lateral meniscus: A histological and computed tomographic study. Knee 2017;24:782-91. [Crossref] [PubMed]
- Branch TP, Siebold R, Freedberg HI, et al. Double-bundle ACL reconstruction demonstrated superior clinical stability to single-bundle ACL reconstruction: a matched-pairs analysis of instrumented tests of tibial anterior translation and internal rotation laxity. Knee Surg Sports Traumatol Arthrosc 2011;19:432-40. [Crossref] [PubMed]
- Siebold R, Branch TP, Freedberg HI, et al. A matched pairs comparison of single- versus double-bundle anterior cruciate ligament reconstructions, clinical results and manual laxity testing. Knee Surg Sports Traumatol Arthrosc 2011;19:S4-S11. [Crossref] [PubMed]
- Siebold R, Dehler C, Ellert T. Prospective randomized comparison of double-bundle versus single-bundle anterior cruciate ligament reconstruction. Arthroscopy 2008;24:137-45. [Crossref] [PubMed]
- Siebold R, Takada T, Feil S, et al. Anatomical "C"-shaped double-bundle versus single-bundle anterior cruciate ligament reconstruction in pre-adolescent children with open growth plates. Knee Surg Sports Traumatol Arthrosc 2016;24:796-806. [Crossref] [PubMed]
Cite this article as: Siebold R, Śmigielski R, Herbort M, Fink C. Anatomical anterior cruciate ligament reconstruction with a flat graft using a new tunnel creation technique. Ann Joint 2019;4:27.