Randomized controlled trial comparing 9 different hip resurfacing designs with a follow-up of 5 years
Introduction
Hip resurfacing arthroplasty (HRA) is employed as an alternative to total hip arthroplasty (THA) in young patients, as it allows preservation of femoral bone stock and resumption of physical activity (1). However, concerns about metal-on-metal (MoM) have led to a decline in hip resurfacing procedures. HRA is a technically challenging procedure. It requires a large amount of experience. We would like to stress the three most important reasons for failure of an HRA. First, as it is a technical challenging procedure, the training and experience of the surgeon is important. The positioning of the acetabular component can only be guided by the surgeon. Second, there are several patient related factors. The available literature reveals a higher rate of complications after MoM HRA in women compared to men (2). Femoral head size has been frequently implicated as a prime factor in the higher rate of complications after HRA. Diagnoses such as developmental hip dysplasia and avascular necrosis have worse results compared to osteoarthritis. The third important reason for failure is the design of the HRA with a small coverage angle as the most important design factor related to edge loading and excessive wear (2).
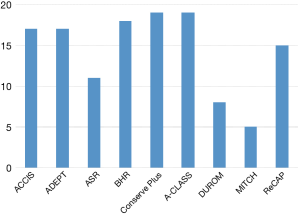
We included nine different hip resurfacing designs on the market in 2007 when the study was started. The designs were the ACCIS (Implantcast GmbH, Buxtehude, Germany), the ADEPT (Finsbury Orthopaedics, Leatherhead, UK), the ASR (DePuy Orthopedics Inc., Warsaw, IN, USA), the BHR (Smith&Nephew Inc., Memphis, TN, USA), the Conserve Plus (Wright Medical Techn. Inc., Arlington, TN, USA), the Conserve Plus AClass (Wright Medical Techn, Inc., Arlington, TN, USA), the DUROM (Zimmer Inc., Warsaw, IN, USA), the MITCH (Stryker Orthopaedics, Mahwah, NJ, USA), and the RECAP (Biomet Orthopedics, Warsaw, IN, USA).
During the course of the study, the ASR, DUROM and MITCH were withdrawn from the market. Despite the withdrawal, the patients who had already received 1 of these 3 designs were kept in the study for follow-up.
Our goal was to investigate these nine different HRA designs and the influence of the abovementioned factors on the outcome of the different designs. Outcome measures included standard clinical and radiological examinations and systemic metal ion levels. An additional research question was whether an experienced surgeon could bypass the possible flaws in an HRA design.
Methods
Patients were randomized to receive 1 of the 9 HRA designs. None of the systems were pre-selected by the manufacturers and all were in clinical use when patients were recruited. Per group, 20 patients would be included to obtain a total population of 180 patients. During the course of the study, the ASR was withdrawn from the market for early failures noted in the Australian Orthopaedic Association Joint Replacement Registry (AOAJRR) (3) whilst the DUROM and MITCH had acetabular fixation problems. As mentioned earlier, patients who had already received 1 of these 3 designs were kept in the study for follow-up.
Randomisation was performed with an online system. Only unilateral HRA were included. Presence of other metal implants (e.g., a total knee arthroplasty) was an exclusion criterium. All the patients had a normal renal function.
Follow-up was conducted at 3, 6, 12, 24 and 60 months. Clinical outcome was evaluated by computing the Harris Hip Score (HHS) (4), which assesses patients’ pain and function, absence of deformity and range of movement of the hips. Patients were grouped according to the Charnley classification [A, single-hip arthropathy; B, contralateral hip arthropathy untreated (B1) or treated (B2); C, multiple arthropathies or medical comorbidities] (4,5). Standing anteroposterior (AP) and lateral radiographs of the pelvis and resurfaced hips were obtained at each follow-up visit.
Metal ion measurements were performed on whole blood, serum and urine samples collected according to a strict protocol (6). This protocol was executed by a single nurse for all the patients. The first 5 mL of blood was discarded to avoid metal contamination from the needle. A second 5 mL of blood was collected using metal-free vacuum tubes for metal ion measurements. Whole blood and serum chromium (Cr) and cobalt (Co) measurements were performed at a single laboratory (University Hospital Ghent) (6).
Results
In September 2011 the inclusion was discontinued. A total of 129 patients had been included by then (Figure 1). Three patients withdrew from the study for personal reasons. Consequently 126 patients remained in the study for the whole 60 months.
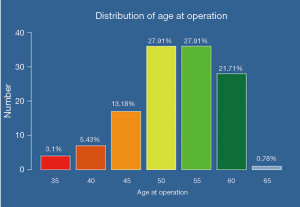
For all patients without exception, the diagnosis was degenerative osteoarthritis. There was no difference in age distribution between the different HRA groups. The mean age was 54.5 years (median 55; range, 37–65 years) (Figure 2).
There was no difference in femoral head size between the different HRA design groups. The mean size was 49.7 mm (median 50 mm; range, 40–60 mm).
There was no statistically significant difference in gender between the HRA groups (P=0.435) but the RECAP group had more female patients while the ADEPT group consisted of mostly male patients. Overall, more men were included (77; 61.1%) than women (49; 38.9%).
Survivorship
Three patients withdrew from the study, and 126 patients remained in the study for the whole 60 months. Two patients passed away almost 4 years after surgery (one female and one male patient), one with a BHR and one with a DUROM, for reasons unrelated to the HRA. When patients received a MoM HRA on the other side, metal ions registration for the study stopped, as the second MoM HRA would influence the results. This was the case in four patients.
Revisions
There were 9 revisions (7%) at a mean of 31.3 months (range, 11–60 months). Six were female patients.
Revision surgery was performed using a ceramic-to-ceramic non-cemented Total Hip Prosthesis (THP).
There were three cases of high metal ions due to excessive wear. One female patient with a RECAP size 50 was revised at 48 months, and another female patient with an ASR, size 46. The last patient with high metal ions was a male with an ACCIS size 54. The HRA was revised at 60 months.
There was one cup loosening of a RECAP in a female patient, size 46, revised at 26 months. One female patient was revised for unclear reasons elsewhere, she had an ACCIS size 50.
We had one low-grade infection in a female with an ASR size 46, revised at 26 months.
Three patients were revised because of suspected metal allergy with low metal ions: a male patient with a BHR at 15 months (size 56), a female patient with a size 46 Conserve Plus revised at 11 months post-op and a male patient with an ADEPT size 54 revised at 15 months post-op.
Lost to follow-up (LTFU)
One male patient with a Conserve Plus was LTFU. There were no data for this patient as of this date. No other patients were LTFU.
Clinical
Of all the remaining implants in the study (n=116), the minimal follow-up was 5 years. At 5 years the mean HHS was 98 (range, 86–100; median 100). We found two outliers (HHS of 90 or less) at the 5-year follow-up appointment. One female patient had an HHS at 2 and at 5 years follow-up of 86. She has an ACCIS prosthesis size 52. The metal ions were always under 4.0 µg/L and the prosthesis is still in situ. At 5 years follow-up a female patient scored 86 with here ACLASS prosthesis size 48. She had slight groin and mid-thigh pain. The prosthesis is still in situ, with normal metal ions.
Metal ions
Whole blood and serum Cr and Co where drawn at the intervals described earlier. We found a high correlation between the whole blood and serum for Cr and Co (r=0.906/0.968; P<0.001). Since Co is ion related to systemic toxicity, we further focus on the Co levels (6,7). There was a significant difference between the preoperative values and postoperative values at every interval (P<0.001). As expected, the Co ion concentration (and Cr) showed a running-in phase until the 1-year follow-up interval. After 1-year follow-up, the Co concentration showed a decrease to a steady-state till the last follow-up interval at 60 months after surgery (7). There were some outliers, most of them female with small femoral heads (RECAP and ASR). Overall Co levels were higher in female patients at 1 year, 2 years and 5 years (P<0.005). Cr levels inversely correlated with head sizes (P<0.01). For Co there was a trend of higher levels with smaller head sizes but no significant difference (P>0.05).
When comparing the metal ion levels of the different HRAs, with exception of the outliers, they all remained below the established safe upper limit for a unilateral HRA at all intervals (Co <4.0 µg/L and Cr <4.6 µg/L) (6). ACCIS and DUROM had the lowest levels compared to other designs in the first 2 years (P<0.01) (Figures 3,4).
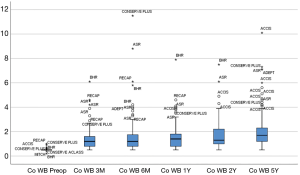
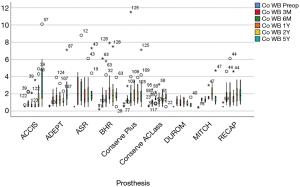
Outliers occurred more often in the RECAP and ASR groups. ACCIS at 1 year (P<0.01) and DUROM at 2 years (P<0.05) had the lowest levels compared to the other resurfacing designs.
The ACCIS design has no traceable Cr and Co ions in the run-in phase because of its TiNb ceramic surface coating preventing Cr and Co release. However, the coating appears to wear off, mostly at the patch area of friction (Figure 5), leading to release of Co and Cr ions. At 5 years of follow-up, besides the revised implants (2 cases), 4 additional ACCIS patients had high metal ions with the prosthesis in situ.

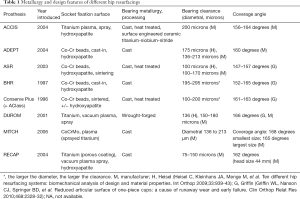
The difference in metal ions between the nine HRA designs is predominantly due to the difference in metallurgy, clearance and coverage angle of the different designs (Table 1).
Full table
The component size and cup inclination or abduction of the acetabular component, determines the amount of coverage of the femoral component.
Radiographic evaluation
In order to accurately evaluate progressive radiographic changes in HRA, specific zones have been established around the femoral component (8). We conducted a review of all the 126 patients by an experienced and unbiased resurfacing surgeon and predominantly we assessed the 9 revision cases.
Component malpositioning [outside of the safe zone, defined as an inclination of the acetabular component of (40–45)°±10°] of an HRA has been associated with high wear (8-10).
When looking at the nine revision cases, we found normal X-rays in five cases. In the three cases with high metal ions we found translucent lines in 3 or more zones in 2 out of 3 cases.
When placing a prosthesis, certainly small sizes (smaller coverage angle), in more than 45° of inclination, the prosthesis will have more chance to fail due to edge loading (10).
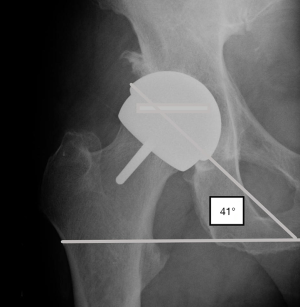
In our study we have 11 ASR, where 2 have been revised. The revision case due to high metal ions, had a small size ASR and was placed in 51° inclination. The mean inclination of the ASR group was 43.6° (range, 40°–53°) When looking at the same ASR group, the biggest size in this group is 58 in a male patient. This prosthesis was placed with an inclination of 41° (Figure 6).
Discussion
This randomized controlled trial (RCT) comparing nine different hip resurfacing designs implanted by a single and experienced hip surgeon shows similar results for all designs, with the exception of a higher number of revisions with the ACCIS (Implantcast GmbH, Buxtehude, Germany), the ASR (DePuy Orthopedics Inc, Warsaw, IN, USA) and the RECAP (Biomet Orthopedics, Warsaw, IN, USA). Clinical and radiographical follow-up with metal ions showed little differences.
A major limitation of this study consisted in the small sample size of the groups. When conducting a RCT, it is important to have a sufficient group size in order to conclude a valid research result. Nevertheless, we were able to bring the RCT to a good end, which is unique in the literature regarding comparison of HRA designs. We showed follow-up data with a minimum of 5 years. Only one patient was LTFU, at 1 year. The blood samples and X-rays are all drawn by one nurse, using a standardized method (5). Last but not least, all patients were operated by one single surgeon with a large experience in hip resurfacing (K De Smet).
The overall revision ratio, at a minimum of 5 years of follow-up, was 7%. In this study comparing nine different HRA designs of which 3 were withdrawn from the market during the course of the study, a <1% per year failure rate was recorded up to 5 years postop. There were 9 revisions (7%) at a mean of 31.3 months (range, 11–60 months). Six were female patients.
Metal ion levels analysis showed similar results for all designs with metal ions remaining below the established upper acceptable limits in all well-functioning HRA (5). Metal allergy leading to adverse inflammatory soft tissue reactions despite low wear and ion levels may occur with any design and was diagnosed in three patients in this study (Conserve Plus, BHR and ADEPT). In the overall population in our clinic, the prevalence of metal allergy is 1 out of 1,300. This is a much smaller rate than the 3 out of 126 in this study. It is not clear whether this is coincidental.
The lowest levels of metal ions were noted with the DUROM prosthesis. This is possibly related to the metallurgy (wrought-forged), the surface finishing, the functional arc of cover of 166° for all sizes and the ideal diametral clearance of 150–180 µm (Table 1) (10). Unfortunately, there was a primary cup stability problem with the DUROM acetabular components which led to their withdrawal from the market.
Overall, the difference between the HRA designs mainly relates to the coverage angle.
When placing a cup, there is more margin for error when the cup has a higher coverage angle. The coverage angle varies between HRA designs (Table 1). Smaller coverage angles will lead to a smaller surface to cover the prosthetic femoral head and will result in edge loading. A prosthesis with a small coverage angle (for example the ASR) can’t withstand steep cup placement in comparison to a prosthesis with a high coverage angle. For example, the DUROM prosthesis has a high coverage angle. There is an articulation arc of 166° for all sizes (10) (Table 1). When placing the acetabular cup of a DUROM at 45° abduction angle, you are giving it a real position of 52° (= effective inclination). When doing so in an ASR prosthesis you can get an effective inclination of 60° or more depending on the size of the cup, so there is a higher risk of edge loading and higher wear (11,12). The difference is even more pronounced in smaller diameters (most female patients).
Other important factors related to HRA outcome are patient-related and surgeon-related. When you combine all risk factors namely smaller component sizes (lower coverage angle) placed in female patients (patient related factor) with hip dysplasia, there is significantly higher risk of failure (12,13) especially when the cup is positioned too vertically. It has repeatedly been highlighted in the literature that inclination angles >45°, were linked to increased requirements for revision surgery (14). An experienced surgeon can bypass these possible obstacles when using an HRA design by placing the cup 45° or less.
However, the ideal patient for an HRA remains a young (<60 years) and active, male patient.
Conclusions
Our goal was to investigate nine different designs of HRA and the influence of these separate designs on the short- and medium-term outcome. This RCT comparing nine different hip resurfacing designs shows similar results for all designs. Clinical and radiographical follow-up with metal ions showed also little differences.
The DUROM prosthesis showed the best results with regard to metal ion levels, probably due to the bigger coverage angle and the metallurgy (Table 1) (10). The ACCIS design had untraceable levels of Cr and Co in the first year due to its TiNbNi coating. In 4 of the 15 remaining patients metal ions were increasing significantly with time. Two of the four outliers in clinical follow-up (HHS) were ACCIS implants and 2 of the 9 revision cases were ACCIS.
Two other important factors are patient related and the surgeon. The difference between the designs is paramount. As shown in Table 1, the coverage angle of every single design is different. When you combine this variable with the smaller sizes (lower coverage angle) placed in female patients (patient related factor), there is a higher chance of failure.
An experienced surgeon can bypass these possible obstacles in an HRA design by placing the cup 45° or less. This was already highlighted in the literature that inclination angles >45°, were linked to increased requirements for revision surgery (12,15).
Overall, we demonstrated very good 5-year clinical, radiographic and metal ions results of the HRA in this RCT comparing nine different designs.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (George Grammatopoulos and Paul E. Beaulé) for the series “Hip Resurfacing for the Young Arthritic Hip” published in Annals of Joint. The article has undergone external peer review.
Conflict of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2019.12.04). The series “Hip Resurfacing for the Young Arthritic Hip” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by institutional ethics board of Jan Palfijn Hospital (No. 225698-07) and informed consent was taken from all the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Quesada MJ, Marker DR, Mont MA. Metal-on-metal hip resurfacing: advantages and disadvantages. J Arthroplasty 2008;23:69-73. [Crossref] [PubMed]
- Haughom BD, Erickson BJ, Hellman MD, et al. Do Complication Rates Differ by Gender After Metal-on-metal Hip Resurfacing Arthroplasty? A Systematic Review. Clin Orthop Relat Res 2015;473:2521-9. [Crossref] [PubMed]
- Wienroth M, McCormack P, Joyce TJ. Precaution, governance and the failure of medical implants: the ASR((TM)) hip in the UK. Life Sci Soc Policy 2014;10:19. [Crossref] [PubMed]
- Harris WH. Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty. An end-result study using a new method of result evaluation. J Bone Joint Surg Am 1969;51:737-55. [Crossref] [PubMed]
- Charnley J, Halley DK. Rate of wear in total hip replacement. Clin Orthop Relat Res 1975;170-9. [PubMed]
- Van Der Straeten C, Van Quickenborne D, De Roest B, et al. Metal ion levels from well-functioning Birmingham Hip Resurfacings decline significantly at ten years. Bone Joint J 2013;95-B:1332-8. [Crossref] [PubMed]
- Bradberry SM, Wilkinson JM, Ferner RE. Systemic toxicity related to metal hip prostheses. Clin Toxicol (Phila) 2014;52:837-47. [Crossref] [PubMed]
- Van Der Straeten C, Calistri A, Grammatopoulos G, et al. Radiographic evaluation of hip resurfacing: the role of x-rays in the diagnosis of a problematic resurfaced hip. Hip Int 2019;1120700019836373 [Epub ahead of print]. [PubMed]
- Van Der Straeten C, Grammatopoulos G, Gill HS, et al. The 2012 Otto Aufranc Award: The interpretation of metal ion levels in unilateral and bilateral hip resurfacing. Clin Orthop Relat Res 2013;471:377-85. [Crossref] [PubMed]
- Heisel C, Kleinhans JA, Menge M, et al. Ten different hip resurfacing systems: biomechanical analysis of design and material properties. Int Orthop 2009;33:939-43. [Crossref] [PubMed]
- Langton DJ, Joyce TJ, Mangat N, et al. Reducing metal ion release following hip resurfacing arthroplasty. Orthop Clin North Am 2011;42:169-80. viii. [Crossref] [PubMed]
- Langton DJ, Joyce TJ, Jameson SS, et al. Adverse reaction to metal debris following hip resurfacing: the influence of component type, orientation and volumetric wear. J Bone Joint Surg Br 2011;93:164-71. [Crossref] [PubMed]
- De Haan R, Pattyn C, Gill HS, et al. Correlation between inclination of the acetabular component and metal ion levels in metal-on-metal hip resurfacing replacement. J Bone Joint Surg Br 2008;90:1291-7. [Crossref] [PubMed]
- de Steiger RN, Graves SE. Orthopaedic registries: the Australian experience. EFORT Open Rev 2019;4:409-15. [Crossref] [PubMed]
- Amstutz HC, Le Duff MJ. Hip resurfacing: history, current status, and future. Hip Int 2015;25:330-8. [Crossref] [PubMed]
Cite this article as: Van Quickenborne D, Van Der Straeten C, De Smet K. Randomized controlled trial comparing 9 different hip resurfacing designs with a follow-up of 5 years. Ann Joint 2019;4:47.