
In vivo kinematics of the knee after discoid lateral subtotal meniscectomy
Introduction
Discoid lateral meniscus (DLM) is a congenital developmental variation of meniscus. The incidence of DLM is not rare especially in Asian populations (1-3). As was reported by the previous studies, there was 3–5% of incidence for Caucasians (2), 15.3% of incidence for Koreans (1) and 16.6% of incidence for Japanese (3). The symptoms caused by symptomatic DLM tears mainly include pain during exercise, loss of motion or typically snapping during knee motion (4). The remaining meniscal tissue can develop into more severe tears if without appropriate treatment (5). Moreover, the meniscal tears are also associated with cartilage degeneration as demonstrated by many clinical or animal studies (6-8).
Recently, although the meniscus-preserving operations including partial meniscectomy with or without meniscal repair were recommended in patients with symptomatic DLM tears (9), the traditional total meniscectomy or meniscal saucerization still remained indispensable due to different tear patterns, tear degrees or neglected severe tears (10). Some studies indicated that the osteoarthritic changes occurred after total meniscectomy or meniscal saucerization in a long-term follow-up (11,12). However, some studies have supported favorable long-term clinical outcomes without obvious radiographic degenerative changes after discoid lateral subtotal meniscectomy (DLSM) in juveniles (13).
Knee kinematics during walking or running, the most common daily activities, are closely related to knee functions and stabilities. Lin et al. (14) found the maximal lateral tibial translation and maximal internal tibial rotation in the knees with DLM injury decreased significantly compared to those with lateral ordinary meniscus injury. They concluded the kinematic features of knees with DLM injury were statistically different from those of healthy knees and knees with lateral ordinary meniscus injury. Harato et al. (15) elucidated three-dimensional knee kinematics in patients with DLM during gait. They found knee excursions in the sagittal and axial plane were significantly smaller on the symptomatic side than on the asymptomatic side in the DLM group. Shoemaker et al. (16) tested the effects of progressive removal of the meniscus on the anterior-posterior force-versus-displacement response of the anterior cruciate ligament (ACL) deficient knee in fresh cadaver specimens. They concluded that the meniscus played an important role in maintaining knee stability and restricting anterior tibial translation. Hosseini et al. (17) demonstrated that the concomitant meniscus tears with ACL injury could affect the knee kinematics in a different way compared to the knees with isolated ACL injuries. Moreover, some studies report that abnormal knee kinematics may be one of the possible reasons for joint degenerations (18,19). The alterations of knee kinematics of the tibiofemoral joint lead to imbalance of cartilage contact pattern, thus resulting in subsequent cartilage degeneration (20).
To the best of our knowledge, few studies have been performed to compare knee kinematics between the affected knees after DLSM and the contralateral normal knees as well as the knees of healthy people during walking and running. The purposes of this study included: (I) the comparison of knee kinematics between the affected knees after DLSM and the contralateral normal knees during walking and running; (II) the comparison of knee kinematics between the affected knees after DLSM and the knees of healthy people during walking.
Methods
Study design
After approval from the institutional review board and obtaining informed consent from all patients, we retrospectively reviewed the medical records from 2018 and 2019 of 21 patients who were previously treated with unilateral primary DLSM for symptomatic DLM tears. Twenty-one healthy volunteers were also recruited for kinematics collection. All subjects were absent of ligament injuries or lower extremity deformities. Before the surgery, the patients with symptomatic DLM tears typically complained of pain localizing to the joint line, swelling, catching, locking, giving way and loss of motion. The physical exam presented with joint effusion, pain in deep flexion or squatting, tenderness with palpation at the lateral joint space or classic presentation of snapping and positive McMurray test. The MRI examination was utilized to evaluate tear location, tear pattern and tear degree. The indications for operation included the aforementioned symptoms and MRI findings. At the last follow-up time, all recruited patients were fully recovered without symptoms and the patients and healthy volunteers were brought back as a group to get the data collected.
Surgical procedure and postoperative rehabilitation
Arthroscopy was performed under general anesthesia. The routine anteromedial (AM) portal and anterolateral (AL) portal for knee arthroscopy were utilized. The standard AL portal was utilized for viewing and the AM portal was used for working. A thorough diagnostic arthroscopy should be implemented to identify all areas of lesions, before any arthroscopic procedures, including discoid lateral meniscal debridement. The knee was placed into flexion and continuous varus stress was applied to the knee, with the leg in the figure-four position, then the lateral compartment was viewed through the AL portal. After determination of DLM tears, the DLSM with the periphery meniscal portion remaining was performed as described by Lee et al. (21). Then the wounds would be closed and the ropivacaine was injected into the wound subcutaneously to relieve pain (Figure 1).
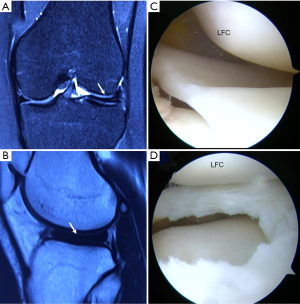
In order to alleviate knee swelling and pain, the ice compress was applied after surgery, immediately. Patients were encouraged to perform ankle pump exercise as early as possible to prevent the formation of lower limb thrombosis. The isometric quadriceps and hamstring contractions, straight and side leg raising exercises were also encouraged to prevent muscle atrophy. The knee extension and flexion exercise were encouraged to increase the range of motion (ROM) of knee. The initiation of low impact exercises can be completed according to the symptoms of patients, then the followings were increasing impact exercises and jogging. The intense physical exercises were not suggested until 3 months after operation.
Kinematics acquisition
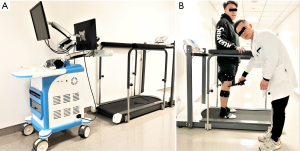
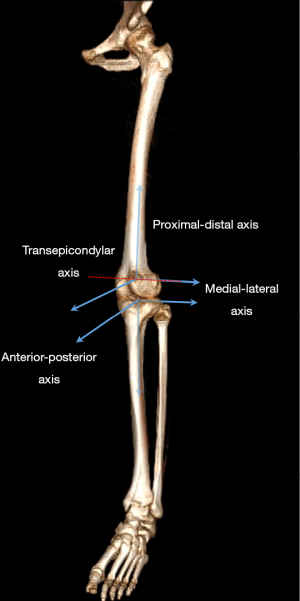
The knee kinematics was acquired by an optical tracking system (Opti-Knee, Innomotion Inc., Shanghai, China) while walking at the speed of 3.0 km/h or running at the speed of 5.0 km/h on a treadmill (Figure 2). The lower limb anatomical bone landmarks were recognized manually, and the infrared-light reflecting rigid bodies were wrapped around the leg as described by a previously published protocol (22). After a 5-minute treadmill warm-up, an integrated stereo-infrared camera at 60 Hz was used to acquire fifteen seconds of kinematics. Simultaneously, an integrated synchronous high-speed camera was utilized to capture the walking or running video for further gait cycle segmentation. Raw data was smoothed by a low-pass filter at a frequency of 6 Hz. The rotational and translational parameters (6 DOF) of knee kinematics were calculated based on the coordinate system of the tibia relative to the femur (Figure 3). The translational parameter was defined as the displacement of the origin of tibial coordinate system relative to the femoral coordinate system, including anterior (+)/posterior translation, proximal (+)/distal translation and medial/lateral (+) translation. Similarly, the rotational parameter was defined as the tibial coordinate system relative to the femoral coordinate system along the anterior-posterior, medial-lateral and proximal-distal axis in the Euler angle sequence, including varus/valgus (+), internal/external (+) rotation, flexion (+)/extension. The ensemble average curve of each DOF was generated by the utilization of MATLAB (2016a; Math-Works Inc.). Cycle segmentation was defined by a kinematic approach mainly including a stance phase and a swing phase (23).
Statistical analysis
The required sample size was computed by a priori power analysis with t-test or Mann-Whitney U test using an α level of 0.05, a power of 0.8 and an effect size of 0.8 by using G*Power software (G*Power 3.1.9.2). The necessary sample size for each group was 21 to achieve a power of 0.8. The data were summarized by the descriptive statistics. All data were presented with means and standard deviations (SD). All data were tested for normality distribution and homogeneity of variance, before statistical analysis was performed. The Shapiro-Wilk test was used to verify the normality distribution, and the Levene statistic was applied to test the homogeneity of variance. Then the unpaired t-test, Welch’s t-test and Mann-Whitney U test were applied according to the result of normality distribution and homogeneity of variance. Statistical analysis was performed with IBM SPSS Statistics 16 (IBM Corporation, NY, USA). A value of P<0.05 was considered to be statistically significant for all tests.
The statistical parametric mapping (1D nonparametric unpaired t-test) was used to evaluate the difference of kinematics between the affected knees and the contralateral normal knees of patients. The difference of kinematics between the affected knees of patients and the knees of healthy people was compared with the same method. The SPM1D package available for MATLAB (v.0.4, http://www.spm1d.org) was used. The SPM1D used Random Field Theory expectations regarding smooth, one-dimensional (random) Gaussian fields to make statistical inferences regarding a set of 1D measurements. More details about SPM1D were published in a previous study (24).
Results
The demographics of patients and healthy volunteers
The demographic data of the patients and healthy volunteers were described in Table 1. Among these 21 patients (21 knees), 11 males and 10 females were included. Among the affected knees, 16 left knees and 5 right knees were treated with DLSM. The average age of patients at operation time was 18 (range, 12–29) years. The average follow-up time was 9 (range, 5–12) months after operation. The average subjective Lysholm score was 91 (range, 82–100).
Full table
Knee kinematics and ROM of 6 DOF analysis
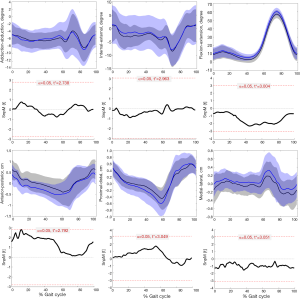
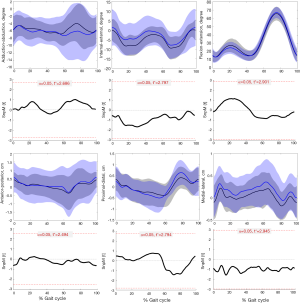
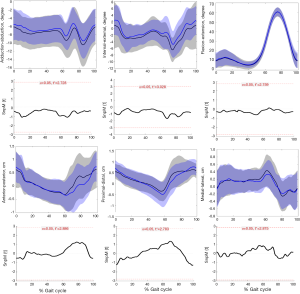
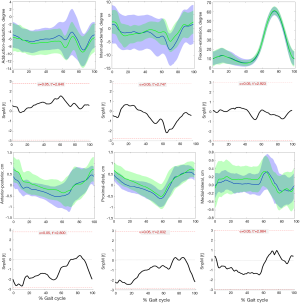
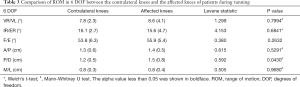
During the entire kinematics cycle, no statistically significant differences were observed in kinematics between the affected knees after DLSM and the contralateral normal knees either in walking (Figure 4) or running (Figure 5) status, as was confirmed by SPM1D analysis. Moreover, both lower extremities of healthy people demonstrated excellent consistence in kinematics (Figure 6). So, the left knees of healthy people were selected for comparison with the affected knees of patients. The kinematics of the affected knees of patients demonstrated no significant differences compared with that of healthy people during walking (Figure 7). The ROM of proximal-distal translation of the affected knees was significantly larger than that of the contralateral normal knees (1.4±0.4 vs. 1.2±0.3 cm; P=0.0418) during walking. However, no statistically significant differences were observed in ROM of the other 5 DOF kinematics between the affected knees and the contralateral normal knees during walking (Table 2). Similarly, The ROM of proximal-distal translation of the affected knees was significantly larger than that of the contralateral normal knees (1.5±0.8 vs. 1.2±0.5 cm; P=0.0430) during running. Nevertheless, no statistically significant differences were observed in ROM of the other 5 DOF kinematics between the affected knees and the contralateral normal knees during running (Table 3). Moreover, there were no significant differences between the affected knees of patients and the knees of healthy people in terms of the ROM of 6 DOF kinematics during walking (Table 4).

Full table
Full table

Full table
Discussion
The first important finding of this study is that there is no significant difference in kinematics between the affected knees and the contralateral knees either in walking or running status during the entire kinematics cycle. The second important finding is that the affected knees exhibited significant larger ROM of proximal-distal translation than the contralateral knees by mean 0.2 cm during walking and 0.3 cm during running.
The postoperative knee kinematics were closely related to the restoration of knee functions and stabilities. Moreover, the abnormal knee kinematics have been reported to be associated with subsequent joint deterioration by many clinical and animal studies (18-20). Shekarforoush et al. (19) elucidated the relations between kinematics alterations and post traumatic osteoarthritic-like changes in sheep injury models. They concluded that the magnitude of the change in the translation vector would be a risk factor for osteoarthritis. Zheng et al. (25) investigated the tibiofemoral skeletal kinematics and cartilage contact arthrokinematics after isolated medial or lateral meniscectomy. They concluded that no consistent difference was observed in skeletal kinematics between the affected knees and the contralateral intact knees, but, significant alterations in the cartilage contact arthrokinematics were demonstrated. As was demonstrated in this study, the affected knees and the contralateral knees presented no significant differences in terms of kinematics. For the affected knees, the average lateral translation of the tibia relative to the femur was greater than that of the contralateral normal knees during waking (Figure 4) or running (Figure 5) through the entire kinematics cycle, even though the difference was not statistically significant. Luczkiewicz et al. (26) investigated the effect of a change in the meniscal cross sectional shape on the biomechanics of a knee joint. They suggested that the medial-lateral translation of the knee joint can be affected by a change in the meniscal shape in the cross-sectional plane. Thus, the alterations in the meniscal shape after DLSM may explain this mild discrepancy of medial-lateral translation between the affected knees and the contralateral knees. However, it should be further verified whether this mild discrepancy could lead to any clinical consequences, such as cartilage degeneration. As for the ROM of kinematics, the affected knees exhibited significant larger ROM of proximal-distal translation than that of the contralateral knees during waking or running. The alterations in the meniscal structure after DLSM may account for this discrepancy.
Currently, the optional surgical treatments for DLM tears included the total meniscectomy or subtotal meniscectomy (27), partial meniscectomy with or without repair (28) and meniscal allograft transplantation (29,30). For those cases with large complex tears, severe meniscal tissue degeneration caused by delayed diagnosis of DLM, then the total meniscectomy or subtotal meniscectomy was inevitable (27,31). The partial meniscectomy was indicated for central portion tears with stable peripheral rim. The aim for partial meniscectomy was to remove the thickened central portion of meniscus and the unstable torn part with a stable peripheral rim more than 6 mm from capsular attachment remaining (28). Although the meniscal tissue preserved strategy for DLM tears was recommended by many surgeons (32,33), the subsequent knee joint degenerations cannot be prevented completely (4). Some studies have also demonstrated favorable long-term clinical outcomes without apparent radiographic degenerative changes after total meniscectomy or subtotal meniscectomy (34-37). Furthermore, the results of this study have demonstrated that the affected knees after DLSM exhibited kinematics symmetry with the contralateral knees during waking or running. And no significant differences were observed between the affected knees and the knees of healthy people in terms of kinematics during walking. Thus, in consideration of postoperative kinematics and clinical outcomes demonstrated by this study, the DLSM still remained to be a valid treatment method for symptomatic DLM tears that met the surgical indications for DLSM.
Some limitations still exist in this study. First, the limited patient sample would not represent the overall situation of kinematics after DLSM. Second, the results demonstrated in this study can only reflect the early stage kinematics after DLSM, with mean follow-up time of 9 months. In addition, this study was limited to knee kinematics testing during walking at the speed of 3.0 km/h and running at the speed of 5.0 km/h. But, the demanding for function of meniscus was different under different states of motion, such as single-leg jump, ascending or descending stairs. Further studies should be performed to evaluate the kinematics after DLSM during different follow-up times as well as under different states of motion. Third, the assessment of cartilage degeneration and joint osteoarthritis-like changes based on radiography or secondary arthroscopy were absent in the current study. Thus, the relation between the kinematics and the potential joint degenerations could not be further illustrated. Finally, it was also acknowledged that the comparison results may be affected by the measurement errors caused by the apparatus and the significant difference of gender, age and height between the recruited patients and healthy people.
Conclusions
In consideration of postoperative kinematics and clinical outcomes demonstrated by this study, the DLSM still remained to be a valid treatment method for symptomatic DLM tears that met the surgical indications for DLSM.
Acknowledgments
We thank the volunteers who participated in this study.
Funding: This work was supported by National Key R&D Program of China (2018YFC1105904), Natural Science Foundation of Jiangsu Province, China (BK20180127), Key Program of NSFC (81730067), Excellent Young Scholars NSFC (81622033), National Science Foundation of China (81802196), Social Development Project of Jiangsu Provincial Science and Technology Department (BE2016609), Six Talent Peaks Project of Jiangsu Province (WSW-061).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2020.01.04). QJ serves as an Editor-in-Chief of Annals of Joint from Mar 2016 to Feb 2021; DS serves as an unpaid Executive Editor-in-Chief of Annals of Joint from Mar 2016 to Feb 2021. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by Medical Ethics Committee of Nanjing Drum Tower Hospital (No. 2019-161-01). Informed consent was taken from all individual participants.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kim SJ, Lee YT, Kim DW. Intraarticular anatomic variants associated with discoid meniscus in Koreans. Clin Orthop Relat Res 1998;202-7. [Crossref] [PubMed]
- Jordan MR. Lateral meniscal variants: evaluation and treatment. J Am Acad Orthop Surg 1996;4:191-200. [Crossref] [PubMed]
- Ikeuchi H. Arthroscopic treatment of the discoid lateral meniscus. Technique and long-term results. Clin Orthop Relat Res 1982;19-28. [PubMed]
- Yamasaki S, Hashimoto Y, Takigami J, et al. Risk factors associated with knee joint degeneration after arthroscopic reshaping for juvenile discoid lateral meniscus. Am J Sports Med 2017;45:570-7. [Crossref] [PubMed]
- Klingele KE, Kocher MS, Hresko MT, et al. Discoid lateral meniscus: prevalence of peripheral rim instability. J Pediatr Orthop 2004;24:79-82. [Crossref] [PubMed]
- Spahn G, Plettenberg H, Hoffmann M, et al. The frequency of cartilage lesions in non-injured knees with symptomatic meniscus tears: results from an arthroscopic and NIR- (near-infrared) spectroscopic investigation. Arch Orthop Trauma Surg 2017;137:837-44. [Crossref] [PubMed]
- Matsubara H, Okazaki K, Takayama Y, et al. Detection of early cartilage deterioration associated with meniscal tear using T1ρ mapping magnetic resonance imaging. BMC Musculoskelet Disord 2015;16:22. [Crossref] [PubMed]
- Steineman BD, LaPrade RF, Santangelo KS, et al. Early osteoarthritis after untreated anterior meniscal root tears: an in vivo animal study. Orthop J Sports Med 2017;5:2325967117702452 [Crossref] [PubMed]
- Adachi N, Ochi M, Uchio Y, et al. Torn discoid lateral meniscus treated using partial central meniscectomy and suture of the peripheral tear. Arthroscopy 2004;20:536-42. [Crossref] [PubMed]
- Jiang D, Luo X, Ao Y, et al. Risk of total/subtotal meniscectomy for respective medial and lateral meniscus injury: correlation with tear type, duration of complaint, age, gender and ACL rupture in 6034 Asian patients. BMC Surg 2017;17:127. [Crossref] [PubMed]
- Lee YS, Teo SH, Ahn JH, et al. Systematic Review of the Long-term Surgical Outcomes of Discoid Lateral Meniscus. Arthroscopy 2017;33:1884-95. [Crossref] [PubMed]
- Räber DA, Friederich NF, Hefti F. Discoid lateral meniscus in children. Long-term follow-up after total meniscectomy. J Bone Joint Surg Am 1998;80:1579-86. [Crossref] [PubMed]
- Stilli S, Marchesini Reggiani L, Marcheggiani Muccioli GM, et al. Arthroscopic treatment for symptomatic discoid lateral meniscus during childhood. Knee Surg Sports Traumatol Arthrosc 2011;19:1337-42. [Crossref] [PubMed]
- Lin Z, Huang W, Ma L, et al. Kinematic features in patients with lateral discoid meniscus injury during walking. Sci Rep 2018;8:5053. [Crossref] [PubMed]
- Harato K, Sakurai A, Kudo Y, et al. Three-dimensional knee kinematics in patients with a discoid lateral meniscus during gait. Knee 2016;23:622-6. [Crossref] [PubMed]
- Shoemaker SC, Markolf KL. The role of the meniscus in the anterior-posterior stability of the loaded anterior cruciate-deficient knee. Effects of partial versus total excision. J Bone Joint Surg Am 1986;68:71-9. [Crossref] [PubMed]
- Hosseini A, Li JS, Gill TJ 4th, et al. Meniscus injuries alter the kinematics of knees with anterior cruciate ligament deficiency. Orthop J Sports Med 2014;2:2325967114547346 [Crossref] [PubMed]
- Andriacchi TP, Favre J. The nature of in vivo mechanical signals that influence cartilage health and progression to knee osteoarthritis. Curr Rheumatol Rep 2014;16:463. [Crossref] [PubMed]
- Shekarforoush M, Beveridge JE, Hart DA, et al. Correlation between translational and rotational kinematic abnormalities and osteoarthritis-like damage in two in vivo sheep injury models. J Biomech 2018;75:67-76. [Crossref] [PubMed]
- Fick JM, P, Ronkainen A, Madden R, et al. Early in situ changes in chondrocyte biomechanical responses due to a partial meniscectomy in the lateral compartment of the mature rabbit knee joint. J Biomech 2016;49:4057-64. [Crossref] [PubMed]
- Lee DH, D'Lima DD, Lee SH. Clinical and radiographic results of partial versus total meniscectomy in patients with symptomatic discoid lateral meniscus: a systematic review and meta-analysis. Orthop Traumatol Surg Res 2019;105:669-75. [Crossref] [PubMed]
- Zhang Y, Huang W, Yao Z, et al. Anterior cruciate ligament injuries alter the kinematics of knees with or without meniscal deficiency. Am J Sports Med 2016;44:3132-9. [Crossref] [PubMed]
- Benedetti MG, Catani F, Leardini A, et al. Data management in gait analysis for clinical applications. Clin Biomech (Bristol, Avon) 1998;13:204-15. [Crossref] [PubMed]
- Pataky TC. One-dimensional statistical parametric mapping in Python. Comput Methods Biomech Biomed Engin 2012;15:295-301. [Crossref] [PubMed]
- Zheng L, Carey R, Thorhauer E, et al. In vivo tibiofemoral skeletal kinematics and cartilage contact arthrokinematics during decline walking after isolated meniscectomy. Med Eng Phys 2018;51:41-8. [Crossref] [PubMed]
- Luczkiewicz P, Daszkiewicz K, Witkowski W, et al. The influence of a change in the meniscus cross-sectional shape on the medio-lateral translation of the knee joint and meniscal extrusion. PLoS One 2018;13:e0193020 [Crossref] [PubMed]
- Lee DH, Kim TH, Kim JM, et al. Results of subtotal/total or partial meniscectomy for discoid lateral meniscus in children. Arthroscopy 2009;25:496-503. [Crossref] [PubMed]
- Ahn JH, Lee SH, Yoo JC, et al. Arthroscopic partial meniscectomy with repair of the peripheral tear for symptomatic discoid lateral meniscus in children: results of minimum 2 years of follow-up. Arthroscopy 2008;24:888-98. [Crossref] [PubMed]
- Yoon KH, Lee SH, Park SY, et al. Meniscus allograft transplantation for discoid lateral meniscus: clinical comparison between discoid lateral meniscus and nondiscoid lateral meniscus. Arthroscopy 2014;30:724-30. [Crossref] [PubMed]
- Kim JM, Bin SI. Meniscal allograft transplantation after total meniscectomy of torn discoid lateral meniscus. Arthroscopy 2006;22:1344-50.e1. [Crossref] [PubMed]
- Yoo WJ, Jang WY, Park MS, et al. Arthroscopic treatment for symptomatic discoid meniscus in children: midterm outcomes and prognostic factors. Arthroscopy 2015;31:2327-34. [Crossref] [PubMed]
- Kramer DE, Micheli LJ. Meniscal tears and discoid meniscus in children: diagnosis and treatment. J Am Acad Orthop Surg 2009;17:698-707. [Crossref] [PubMed]
- Kushare I, Klingele K, Samora W. Discoid meniscus: diagnosis and management. Orthop Clin North Am 2015;46:533-40. [Crossref] [PubMed]
- Habata T, Uematsu K, Kasanami R, et al. Long-term clinical and radiographic follow-up of total resection for discoid lateral meniscus. Arthroscopy 2006;22:1339-43. [Crossref] [PubMed]
- Okazaki K, Miura H, Matsuda S, et al. Arthroscopic resection of the discoid lateral meniscus: long-term follow-up for 16 years. Arthroscopy 2006;22:967-71. [Crossref] [PubMed]
- Aichroth PM, Patel DV, Marx CL. Congenital discoid lateral meniscus in children. A follow-up study and evolution of management. J Bone Joint Surg Br 1991;73:932-6. [Crossref] [PubMed]
- Washington ER 3rd, Root L, Liener UC. Discoid lateral meniscus in children. Long-term follow-up after excision. J Bone Joint Surg Am 1995;77:1357-61. [Crossref] [PubMed]
Cite this article as: Yan W, Xu X, Xu Q, Chen D, Xu Z, Dai J, Jiang Q, Shi D. In vivo kinematics of the knee after discoid lateral subtotal meniscectomy. Ann Joint 2020;5:11.


